Glioblastoma cells require glutamate dehydrogenase to survive impairments of glucose metabolism or Akt signaling
- PMID: 19826036
- PMCID: PMC2764330
- DOI: 10.1158/0008-5472.CAN-09-2266
Glioblastoma cells require glutamate dehydrogenase to survive impairments of glucose metabolism or Akt signaling
Erratum in
- Cancer Res. 2010 Feb 1;70(3):1275. Bachoo, Robert G [corrected to Bachoo, Robert M]
Abstract
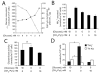
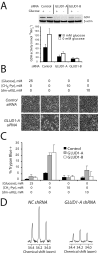
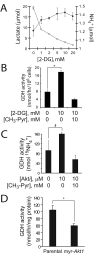
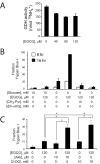
Oncogenes influence nutrient metabolism and nutrient dependence. The oncogene c-Myc stimulates glutamine metabolism and renders cells dependent on glutamine to sustain viability ("glutamine addiction"), suggesting that treatments targeting glutamine metabolism might selectively kill c-Myc-transformed tumor cells. However, many current or proposed cancer therapies interfere with the metabolism of glucose, not glutamine. Here, we studied how c-Myc-transformed cells maintained viability when glucose metabolism was impaired. In SF188 glioblastoma cells, glucose deprivation did not affect net glutamine utilization but elicited a switch in the pathways used to deliver glutamine carbon to the tricarboxylic acid cycle, with a large increase in the activity of glutamate dehydrogenase (GDH). The effect on GDH resulted from the loss of glycolysis because it could be mimicked with the glycolytic inhibitor 2-deoxyglucose and reversed with a pyruvate analogue. Furthermore, inhibition of Akt signaling, which facilitates glycolysis, increased GDH activity whereas overexpression of Akt suppressed it, suggesting that Akt indirectly regulates GDH through its effects on glucose metabolism. Suppression of GDH activity with RNA interference or an inhibitor showed that the enzyme is dispensable in cells able to metabolize glucose but is required for cells to survive impairments of glycolysis brought about by glucose deprivation, 2-deoxyglucose, or Akt inhibition. Thus, inhibition of GDH converted these glutamine-addicted cells to glucose-addicted cells. The findings emphasize the integration of glucose metabolism, glutamine metabolism, and oncogenic signaling in glioblastoma cells and suggest that exploiting compensatory pathways of glutamine metabolism can improve the efficacy of cancer treatments that impair glucose utilization.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

