Comparison of immunopathology and locomotor recovery in C57BL/6, BUB/BnJ, and NOD-SCID mice after contusion spinal cord injury
- PMID: 19831737
- PMCID: PMC2834443
- DOI: 10.1089/neu.2009.0930
Comparison of immunopathology and locomotor recovery in C57BL/6, BUB/BnJ, and NOD-SCID mice after contusion spinal cord injury
Abstract
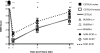
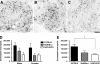
Studies of cell transplantation therapeutics in animal models of traumatic spinal cord injury (SCI) are often hampered by partial or complete rejection of the graft by the host. Pharmacological immunosuppression is rarely sufficient to prevent rejection. Further, the immunological niche created by both the host immune response and immunosuppressant drugs could hypothetically influence the proliferation, differentiation, and fate of transplanted progenitor/stem cells. To avoid these confounds, we have previously used the constitutively immunodeficient non-obese diabetic severe combined immunodeficient (NOD-SCID) mouse as a model for transplantation studies following SCI. In the current study, we compare behavioral and histological recovery in NOD-SCID, C57BL/6, and BUB/BnJ mice of both sexes to better facilitate interpretation of data from studies using NOD-SCID mice. Of the strains examined, NOD-SCID mice exhibited the greatest locomotor recovery in the open field; no sex differences were detected in locomotor recovery in any of the strains. Stereologic estimation of the number of infiltrated neutrophils showed more cells in C57BL/6 mice than NOD-SCID mice, with BUB/BnJ mice having an intermediate number. The volume of macrophages/microglia did not differ between strains or sexes, though more rostral-caudal spreading was observed in C57BL/6 and BUB/BnJ than NOD-SCID mice. No significant differences were detected in lesion volume. Taken together these findings demonstrate that relative to other strains, NOD-SCID mice have both similar primary lesion volume and cellular inflammatory parameters after SCI, and support the applicability of the model for neurotransplantation studies.
Figures



Similar articles
-
Deficiency in complement C1q improves histological and functional locomotor outcome after spinal cord injury.J Neurosci. 2008 Dec 17;28(51):13876-88. doi: 10.1523/JNEUROSCI.2823-08.2008. J Neurosci. 2008. PMID: 19091977 Free PMC article.
-
Traumatic spinal cord injury in mice with human immune systems.Exp Neurol. 2015 Sep;271:432-44. doi: 10.1016/j.expneurol.2015.07.011. Epub 2015 Jul 17. Exp Neurol. 2015. PMID: 26193167 Free PMC article.
-
Human neural stem cells differentiate and promote locomotor recovery in an early chronic spinal cord injury NOD-scid mouse model.PLoS One. 2010 Aug 18;5(8):e12272. doi: 10.1371/journal.pone.0012272. PLoS One. 2010. PMID: 20806064 Free PMC article.
-
Improved regeneration after spinal cord injury in mice lacking functional T- and B-lymphocytes.Exp Neurol. 2012 Oct;237(2):274-85. doi: 10.1016/j.expneurol.2012.07.016. Epub 2012 Jul 31. Exp Neurol. 2012. PMID: 22868200
-
[Application of NOD/SCID mice in research of experimental hematology - review].Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2008 Aug;16(4):964-8. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2008. PMID: 18718101 Review. Chinese.
Cited by
-
Enhanced oxidative phosphorylation, re-organized intracellular signaling, and epigenetic de-silencing as revealed by oligodendrocyte translatome analysis after contusive spinal cord injury.Sci Rep. 2023 Dec 1;13(1):21254. doi: 10.1038/s41598-023-48425-6. Sci Rep. 2023. PMID: 38040794 Free PMC article.
-
miR-155 Deletion in Mice Overcomes Neuron-Intrinsic and Neuron-Extrinsic Barriers to Spinal Cord Repair.J Neurosci. 2016 Aug 10;36(32):8516-32. doi: 10.1523/JNEUROSCI.0735-16.2016. J Neurosci. 2016. PMID: 27511021 Free PMC article.
-
The Effects of Icariin on Enhancing Motor Recovery Through Attenuating Pro-inflammatory Factors and Oxidative Stress via Mitochondrial Apoptotic Pathway in the Mice Model of Spinal Cord Injury.Front Physiol. 2018 Nov 16;9:1617. doi: 10.3389/fphys.2018.01617. eCollection 2018. Front Physiol. 2018. PMID: 30505282 Free PMC article.
-
Achieving stable human stem cell engraftment and survival in the CNS: is the future of regenerative medicine immunodeficient?Regen Med. 2011 May;6(3):367-406. doi: 10.2217/rme.11.22. Regen Med. 2011. PMID: 21548741 Free PMC article.
-
Restoring mitochondrial cardiolipin homeostasis reduces cell death and promotes recovery after spinal cord injury.Cell Death Dis. 2022 Dec 20;13(12):1058. doi: 10.1038/s41419-022-05369-5. Cell Death Dis. 2022. PMID: 36539405 Free PMC article.
References
-
- Abe K. Endo Y. Nakazawa N. Kanno K. Okubo M. Hoshino T. Fujita T. Unique phenotypes of C1s deficiency and abnormality caused by two compound heterozygosities in a Japanese family. J. Immunol. 2009;182:1681–1688. - PubMed
-
- Alberti K. Davey R.E. Onishi K. George S. Salchert K. Seib F.P. Bornhauser M. Pompe T. Nagy A. Werner C. Zandstra P.W. Functional immobilization of signaling proteins enables control of stem cell fate. Nat. Methods. 2008;5:645–650. - PubMed
-
- Anderson K.D. Targeting recovery: priorities of the spinal cord-injured population. J. Neurotrauma. 2004;21:1371–1383. - PubMed
-
- Ankeny D.P. Lucin K.M. Sanders V.M. McGaughy V.M. Popovich P.G. Spinal cord injury triggers systemic autoimmunity: evidence for chronic B lymphocyte activation and lupus-like autoantibody synthesis. J. Neurochem. 2006;99:1073–1087. - PubMed
-
- Armstrong R.J. Harrower T.P. Hurelbrink C.B. McLaughin M. Ratcliffe E.L. Tyers P. Richards A. Dunnett S.B. Rosser A.E. Barker R.A. Porcine neural xenografts in the immunocompetent rat: immune response following grafting of expanded neural precursor cells. Neuroscience. 2001;106:201–216. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

