The endocrine control of reproduction in Nereidae: a new multi-hormonal model with implications for their functional role in a changing environment
- PMID: 19833648
- PMCID: PMC2781847
- DOI: 10.1098/rstb.2009.0127
The endocrine control of reproduction in Nereidae: a new multi-hormonal model with implications for their functional role in a changing environment
Abstract
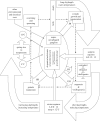
Nereidae are vital to the functioning of estuarine ecosystems and are major components in the diets of over-wintering birds and commercial fish. They use environmental cues to synchronize reproduction. Photoperiod is the proximate cue, initiating vitellogenesis in a temperature-compensated process. The prevailing paradigm in Nereidae is of a single 'juvenile' hormone controlling growth and reproduction. However, a new multi-hormone model is presented here that integrates the environmental and endocrine control of reproduction. This is supported by evidence from in vitro bioassays. The juvenile hormone is shown to be heat stable and cross reactive between species. In addition, a second neuro-hormone, identified here as a gonadotrophic hormone, is shown to be present in mature females and is found to promote oocyte growth. Furthermore, dopamine and melatonin appear to switch off the juvenile hormone while serotonin and oxytocin promote oocyte growth. Global warming is likely to uncouple the phase relationship between temperature and photoperiod, with significant consequences for Nereidae that use photoperiod to cue reproduction during the winter in northern latitudes. Genotypic adaptation of the photoperiodic response may be possible, but significant impacts on fecundity, spawning success and recruitment are likely in response to short-term extreme events. Endocrine-disrupting chemicals may also impact on putative steroid hormone pathways in Nereidae with similar consequences. These impacts may have significant implications for the functional role of Nereidae and highlight the importance of comparative endocrinology studies in these and other invertebrates.
Figures
Similar articles
-
Factors influencing spawning and pairing in the scale worm Harmothoe imbricata (Annelida: Polychaeta).Biol Bull. 2000 Aug;199(1):50-8. doi: 10.2307/1542706. Biol Bull. 2000. PMID: 10975642
-
Mysid crustaceans as potential test organisms for the evaluation of environmental endocrine disruption: a review.Environ Toxicol Chem. 2004 May;23(5):1219-34. doi: 10.1897/03-332. Environ Toxicol Chem. 2004. PMID: 15180373 Review.
-
Ambient temperature effects on photo induced gonadal cycles and hormonal secretion patterns in Great Tits from three different breeding latitudes.Horm Behav. 2008 Jun;54(1):60-8. doi: 10.1016/j.yhbeh.2008.01.015. Epub 2008 Mar 10. Horm Behav. 2008. PMID: 18402961
-
Climate change and seasonal reproduction in mammals.Philos Trans R Soc Lond B Biol Sci. 2009 Nov 27;364(1534):3331-40. doi: 10.1098/rstb.2009.0140. Philos Trans R Soc Lond B Biol Sci. 2009. PMID: 19833645 Free PMC article.
-
Broodstock management and hormonal manipulations of fish reproduction.Gen Comp Endocrinol. 2010 Feb 1;165(3):516-34. doi: 10.1016/j.ygcen.2009.03.007. Epub 2009 Mar 24. Gen Comp Endocrinol. 2010. PMID: 19318108 Review.
Cited by
-
On the role of the proventricle region in reproduction and regeneration in Typosyllis antoni (Annelida: Syllidae).BMC Evol Biol. 2016 Oct 4;16(1):196. doi: 10.1186/s12862-016-0770-5. BMC Evol Biol. 2016. PMID: 27716025 Free PMC article.
-
The cost and payout of age on germline regeneration and sexual maturation in Platynereis dumerilii.bioRxiv [Preprint]. 2024 Jan 22:2024.01.22.576726. doi: 10.1101/2024.01.22.576726. bioRxiv. 2024. Update in: Dev Biol. 2024 Sep;513:33-49. doi: 10.1016/j.ydbio.2024.05.013. PMID: 38328233 Free PMC article. Updated. Preprint.
-
Sex-specific differential gene expression during stolonization in the branching syllid Ramisyllis kingghidorahi (Annelida, Syllidae).BMC Genomics. 2025 Apr 25;26(1):405. doi: 10.1186/s12864-025-11587-w. BMC Genomics. 2025. PMID: 40275122 Free PMC article.
-
Impacts of climate change and environmental factors on reproduction and development in wildlife.Philos Trans R Soc Lond B Biol Sci. 2009 Nov 27;364(1534):3313-9. doi: 10.1098/rstb.2009.0175. Philos Trans R Soc Lond B Biol Sci. 2009. PMID: 19833643 Free PMC article.
-
Delegating Sex: Differential Gene Expression in Stolonizing Syllids Uncovers the Hormonal Control of Reproduction.Genome Biol Evol. 2019 Jan 1;11(1):295-318. doi: 10.1093/gbe/evy265. Genome Biol Evol. 2019. PMID: 30535381 Free PMC article.
References
-
- Ambrose W. G.1986Estimate of removal rate of Nereis virens (Polychaeta: Nereidae) from an intertidal mudflat by gulls (Larus spp.). Mar. Biol. 90, 243–247 (doi:10.1007/BF00569134) - DOI
-
- Andries J. C.2001Endocrine and environmental control of reproduction in Polychaeta. Can. J. Zool. 79, 254–270 (doi:10.1139/cjz-79-2-254) - DOI
-
- Arendt D., Tessmar-Raible K., Snyman H., Dorresteijn A. W., Wittbrodt J.2004Ciliary photoreceptors with a vertebrate-type opsin in an invertebrate brain. Science 306, 869–871 (doi:10.1126/science.1099955) - DOI - PubMed
-
- Arnoult F., Vernet G.1996Influence of melatonin on gonad maturation in Lineus lacteus (Heteronemertini, Lineidae). J. Exp. Zool. 274, 138–143 (doi:10.1002/(SICI)1097-010X(19960201)274:2<138::AID-JEZ7>3.0.CO;2-Q) - DOI
-
- Baduini C. L., Hyrenbach K. D., Coyle K. O., Pinchuk A., Mendenhall V., Hunt G. L.2001Mass mortality of Short-tailed Shearwaters in the south-eastern Bering Sea during summer 1997. Fisheries Oceanogr. 10, 117–130 (doi:10.1046/j.1365-2419.2001.00156.x) - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

