The olfactory nerve has a role in the body temperature and brain cytokine responses to influenza virus
- PMID: 19836444
- PMCID: PMC2818451
- DOI: 10.1016/j.bbi.2009.10.007
The olfactory nerve has a role in the body temperature and brain cytokine responses to influenza virus
Abstract
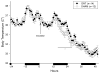
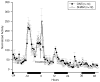
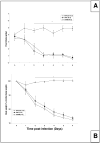
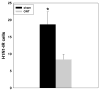
Mouse-adapted human influenza virus is detectable in the olfactory bulbs of mice within hours after intranasal challenge and is associated with enhanced local cytokine mRNA and protein levels. To determine whether signals from the olfactory nerve influence the unfolding of the acute phase response (APR), we surgically transected the olfactory nerve in mice prior to influenza infection. We then compared the responses of olfactory-nerve-transected (ONT) mice to those recorded in sham-operated control mice using measurements of body temperature, food intake, body weight, locomotor activity and immunohistochemistry for cytokines and the viral antigen, H1N1. ONT did not change baseline body temperature (Tb); however, the onset of virus-induced hypothermia was delayed for about 13 h in the ONT mice. Locomotor activity, food intake and body weights of the two groups were similar. At 15 h post-challenge fewer viral antigen-immunoreactive (IR) cells were observed in the olfactory bulb (OB) of ONT mice compared to sham controls. The number of tumor necrosis factor alpha (TNFalpha)- and interleukin 1beta (IL1beta)-IR cells in ONT mice was also reduced in the OB and other interconnected regions in the brain compared to sham controls. These results suggest that the olfactory nerve pathway is important for the initial pathogenesis of the influenza-induced APR.
Copyright 2009 Elsevier Inc. All rights reserved.
Figures


References
-
- Alt JA, Bohnet S, Taishi P, Durika D, Obal F, Traynor T, Majde JA, Krueger JM. Influenza virus-induced glucocorticoid and hypothalamic and lung cytokine mRNA responses in dwarf lit/lit mice. Brain Behav Immun. 2007;21:60–67. - PubMed
-
- Barnett EM, Perlman S. The olfactory nerve and not the trigeminal nerve is the major site of CNS entry for mouse hepatitis virus, strain JHM. Virology. 1993;194:185–191. - PubMed
-
- Bluthé RM, Layé S, Michaud B, Combe C, Dantzer R, Parnet P. Role of interleukin-1beta and tumour necrosis factor-alpha in lipopolysaccharide-induced sickness behaviour: a study with interleukin-1 type I receptor-deficient mice. Eur J Neurosci. 2000;12:4447–4456. - PubMed
-
- Bugajski AJ, Gil K, Ziomber A, Zurowski D, Zaraska W, Thor PJ. Effect of long-term vagal stimulation on food intake and body weight during diet induced obesity in rats. J Physiol Pharmacol. 2007;58(Suppl 1):5–12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

