Small RNA-dependent expression of secondary metabolism is controlled by Krebs cycle function in Pseudomonas fluorescens
- PMID: 19840935
- PMCID: PMC2787360
- DOI: 10.1074/jbc.M109.052571
Small RNA-dependent expression of secondary metabolism is controlled by Krebs cycle function in Pseudomonas fluorescens
Abstract
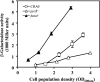
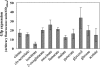
Pseudomonas fluorescens CHA0, an antagonist of phytopathogenic fungi in the rhizosphere of crop plants, elaborates and excretes several secondary metabolites with antibiotic properties. Their synthesis depends on three small RNAs (RsmX, RsmY, and RsmZ), whose expression is positively controlled by the GacS-GacA two-component system at high cell population densities. To find regulatory links between primary and secondary metabolism in P. fluorescens and in the related species Pseudomonas aeruginosa, we searched for null mutations that affected central carbon metabolism as well as the expression of rsmY-gfp and rsmZ-gfp reporter constructs but without slowing down the growth rate in rich media. Mutation in the pycAB genes (for pyruvate carboxylase) led to down-regulation of rsmXYZ and secondary metabolism, whereas mutation in fumA (for a fumarase isoenzyme) resulted in up-regulation of the three small RNAs and secondary metabolism in the absence of detectable nutrient limitation. These effects required the GacS sensor kinase but not the accessory sensors RetS and LadS. An analysis of intracellular metabolites in P. fluorescens revealed a strong positive correlation between small RNA expression and the pools of 2-oxoglutarate, succinate, and fumarate. We conclude that Krebs cycle intermediates (already known to control GacA-dependent virulence factors in P. aeruginosa) exert a critical trigger function in secondary metabolism via the expression of GacA-dependent small RNAs.
Figures





Similar articles
-
RsmY, a small regulatory RNA, is required in concert with RsmZ for GacA-dependent expression of biocontrol traits in Pseudomonas fluorescens CHA0.Mol Microbiol. 2003 Nov;50(4):1361-79. doi: 10.1046/j.1365-2958.2003.03774.x. Mol Microbiol. 2003. PMID: 14622422
-
Temperature-responsive sensing regulates biocontrol factor expression in Pseudomonas fluorescens CHA0.ISME J. 2009 Aug;3(8):955-65. doi: 10.1038/ismej.2009.42. Epub 2009 May 7. ISME J. 2009. PMID: 19421236
-
Three small RNAs jointly ensure secondary metabolism and biocontrol in Pseudomonas fluorescens CHA0.Proc Natl Acad Sci U S A. 2005 Nov 22;102(47):17136-41. doi: 10.1073/pnas.0505673102. Epub 2005 Nov 14. Proc Natl Acad Sci U S A. 2005. PMID: 16286659 Free PMC article.
-
Signal transduction in plant-beneficial rhizobacteria with biocontrol properties.Antonie Van Leeuwenhoek. 2002 Aug;81(1-4):385-95. doi: 10.1023/a:1020549019981. Antonie Van Leeuwenhoek. 2002. PMID: 12448737 Review.
-
Pseudomonas fluorescens orchestrates a fine metabolic-balancing act to counter aluminium toxicity.Environ Microbiol. 2010 Jun;12(6):1384-90. doi: 10.1111/j.1462-2920.2010.02200.x. Epub 2010 Mar 25. Environ Microbiol. 2010. PMID: 20353438 Review.
Cited by
-
Regulation of bacterial virulence by Csr (Rsm) systems.Microbiol Mol Biol Rev. 2015 Jun;79(2):193-224. doi: 10.1128/MMBR.00052-14. Microbiol Mol Biol Rev. 2015. PMID: 25833324 Free PMC article. Review.
-
Bile signalling promotes chronic respiratory infections and antibiotic tolerance.Sci Rep. 2016 Jul 19;6:29768. doi: 10.1038/srep29768. Sci Rep. 2016. PMID: 27432520 Free PMC article.
-
Computational prediction of the Crc regulon identifies genus-wide and species-specific targets of catabolite repression control in Pseudomonas bacteria.BMC Microbiol. 2010 Nov 25;10:300. doi: 10.1186/1471-2180-10-300. BMC Microbiol. 2010. PMID: 21108798 Free PMC article.
-
Crp induces switching of the CsrB and CsrC RNAs in Yersinia pseudotuberculosis and links nutritional status to virulence.Front Cell Infect Microbiol. 2012 Dec 17;2:158. doi: 10.3389/fcimb.2012.00158. eCollection 2012. Front Cell Infect Microbiol. 2012. PMID: 23251905 Free PMC article.
-
The expression of stlA in Photorhabdus luminescens is controlled by nutrient limitation.PLoS One. 2013 Nov 22;8(11):e82152. doi: 10.1371/journal.pone.0082152. eCollection 2013. PLoS One. 2013. PMID: 24278476 Free PMC article.
References
-
- Bibb M. J. (2005) Curr. Opin. Microbiol. 8, 208–215 - PubMed
-
- Yu J. H., Keller N. (2005) Annu. Rev. Phytopathol. 43, 437–458 - PubMed
-
- Vining L. C. (1990) Annu. Rev. Microbiol. 44, 395–427 - PubMed
-
- Haas D., Keel C. (2003) Annu. Rev. Phytopathol. 41, 117–153 - PubMed
-
- Demain A. (1986) Pure Appl. Chem. 58, 219–226
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

