Synthesis of Homopolymers and Copolymers Containing an Active Ester of Acrylic Acid by RAFT: Scaffolds for Controlling Polyvalent Ligand Display
- PMID: 19855852
- PMCID: PMC2765779
- DOI: 10.1002/pola.23031
Synthesis of Homopolymers and Copolymers Containing an Active Ester of Acrylic Acid by RAFT: Scaffolds for Controlling Polyvalent Ligand Display
Abstract
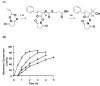
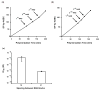
We describe the synthesis of activated homopolymers and copolymers of controlled molecular weight based on the controlled radical polymerization of N-acryloyloxysuccinimide (NAS) by reversible addition fragmentation chain transfer (RAFT). We synthesized activated homopolymers in a range of molecular weights with polydispersities between 1 and 1.2. The attachment of an inhibitory peptide to the activated polymer backbone yielded a potent controlled molecular weight polyvalent inhibitor of anthrax toxin. To provide greater control over the placement of the peptides along the polymer backbone, we also used a semi-batch copolymerization method to synthesize copolymers of NAS and acrylamide (AAm). This approach enabled the synthesis of copolymers with control over the placement of peptide-reactive NAS monomers along an inert backbone; subsequent functionalization of NAS with peptide yielded well-defined polyvalent anthrax toxin inhibitors that differed in their potencies. These strategies for controlling molecular weight, ligand density, and ligand placement will be broadly applicable for designing potent polyvalent inhibitors for a variety of pathogens and toxins, and for elucidating structure-activity relationships in these systems.
Figures

Similar articles
-
Synthesis of copolymers containing an active ester of methacrylic acid by RAFT: controlled molecular weight scaffolds for biofunctionalization.Biomacromolecules. 2006 May;7(5):1665-70. doi: 10.1021/bm060098v. Biomacromolecules. 2006. PMID: 16677052
-
Synthesis of polyvalent inhibitors of controlled molecular weight: structure-activity relationship for inhibitors of anthrax toxin.Biomacromolecules. 2006 Jul;7(7):2082-5. doi: 10.1021/bm060210p. Biomacromolecules. 2006. PMID: 16827573
-
Design of monodisperse and well-defined polypeptide-based polyvalent inhibitors of anthrax toxin.Angew Chem Int Ed Engl. 2014 Jul 28;53(31):8037-40. doi: 10.1002/anie.201400870. Epub 2014 Apr 6. Angew Chem Int Ed Engl. 2014. PMID: 24706570 Free PMC article.
-
Using RAFT Polymerization Methodologies to Create Branched and Nanogel-Type Copolymers.Materials (Basel). 2024 Apr 23;17(9):1947. doi: 10.3390/ma17091947. Materials (Basel). 2024. PMID: 38730753 Free PMC article. Review.
-
Polyhomologation. A living C1 polymerization.Acc Chem Res. 2010 Nov 16;43(11):1420-33. doi: 10.1021/ar100062a. Epub 2010 Sep 8. Acc Chem Res. 2010. PMID: 20825177 Review.
Cited by
-
Self-Healing Thermosensitive Hydrogel for Sustained Release of Dexamethasone for Ocular Therapy.ACS Polym Au. 2022 Nov 3;3(1):118-131. doi: 10.1021/acspolymersau.2c00038. eCollection 2023 Feb 8. ACS Polym Au. 2022. PMID: 36785837 Free PMC article.
-
Polymeric drugs: Advances in the development of pharmacologically active polymers.J Control Release. 2015 Dec 10;219:369-382. doi: 10.1016/j.jconrel.2015.09.043. Epub 2015 Sep 26. J Control Release. 2015. PMID: 26410809 Free PMC article. Review.
-
Designing inhibitors of anthrax toxin.Expert Opin Drug Discov. 2014 Mar;9(3):299-318. doi: 10.1517/17460441.2014.877884. Epub 2014 Jan 22. Expert Opin Drug Discov. 2014. PMID: 24447197 Free PMC article. Review.
-
An overview of investigational toxin-directed therapies for the adjunctive management of Bacillus anthracis infection and sepsis.Expert Opin Investig Drugs. 2015;24(7):851-65. doi: 10.1517/13543784.2015.1041587. Epub 2015 Apr 28. Expert Opin Investig Drugs. 2015. PMID: 25920540 Free PMC article. Review.
-
Multivalent Inhibitors of Channel-Forming Bacterial Toxins.Curr Top Microbiol Immunol. 2017;406:199-227. doi: 10.1007/82_2016_20. Curr Top Microbiol Immunol. 2017. PMID: 27469304 Free PMC article. Review.
References
-
- Mammen M, Choi SK, Whitesides GM. Angew Chem Intl Ed. 1998;37:2755–2794. - PubMed
-
- Kiessling LL, Strong LE, Gestwicki JE. Annu Rep Med Chem. 2000;35:321–330.
-
- Mulder A, Huskens J, Reinhoudt DN. Org Biomol Chem. 2004;2:3409–3424. - PubMed
-
- Kane RS. AIChE J. 2006;52:3638 – 3644.
-
- Mammen M, Dahmann G, Whitesides GMJ. Med Chem. 1995;38:4179–4190. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
