Glucose promotes the production of interleukine-1beta and cyclooxygenase-2 in mesangial cells via enhanced (Pro)renin receptor expression
- PMID: 19861503
- PMCID: PMC2795703
- DOI: 10.1210/en.2009-0442
Glucose promotes the production of interleukine-1beta and cyclooxygenase-2 in mesangial cells via enhanced (Pro)renin receptor expression
Abstract
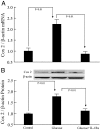
(Pro)renin receptor (PRR) is present in renal glomeruli, and its expression is up-regulated in diabetes. Similarly, renal inflammation is increased in the presence of hyperglycemia. The linkage between PRR and renal inflammation is not well established. We hypothesized that glucose-induced up-regulation of PRR leads to increased production of the proinflammatory factors IL-1beta and cyclooxygenase-2 (COX-2). Studies were conducted in rat mesangial cells (RMCs) exposed to 30 mm D-glucose for 2 wk followed by PRR small interfering RNA knockdown, IL-1 receptor blockade with IL-1 receptor antagonist or angiotensin II type 1 receptor blockade with valsartan. The results showed that D-glucose treatment up-regulates prorenin, renin, angiotensin II, PRR, IL-1beta, and COX-2 mRNA and protein expression and increases phosphorylation of ERK1/2, c-Jun N-terminal kinase, c-Jun, and nuclear factor-kappaB (NF-kappaB) p65 (serine 276,468 and 536), respectively. PRR small interfering RNA attenuated PRR, IL-1beta, and COX-2 mRNA and protein expressions and significantly decreased angiotensin II production and phosphorylation of ERK1/2 and NF-kappaB p65 associated with high glucose exposure. Similarly, IL-1 receptor antagonist significantly reduced COX-2 mRNA and protein expression induced by high glucose. COX-2 inhibition reduced high-glucose-induced PRR expression. We conclude that glucose induces the up-regulation of PRR and its ligands prorenin and renin, leading to increased IL-1beta and COX-2 production via the angiotensin II-dependent pathway. It is also possible that PRR could enhance the production of these inflammatory cytokines through direct stimulation of ERK1/2-NF-kappaB signaling cascade.
Figures


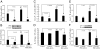


References
-
- Atiyeh BA, Arant Jr BS, Henrich WL, Seikaly MG 1995 In vitro production of angiotensin II by isolated glomeruli. Am J Physiol 268:F266–F272 - PubMed
-
- Becker BN, Yasuda T, Kondo S, Vaikunth S, Homma T, Harris RC 1998 Mechanical stretch/relaxation stimulates a cellular renin-angiotensin system in cultured rat mesangial cells. Exp Nephrol 6:57–66 - PubMed
-
- Andrade MC, Quinto BM, Carmona AK, Ribas OS, Boim MA, Schor N, Casarini DE 1998 Purification and characterization of angiotensin I-converting enzymes from mesangial cells in culture. J Hypertens 16:2063–2074 - PubMed
-
- Singh R, Singh AK, Alavi N, Leehey DJ 2003 Mechanism of increased angiotensin II levels in glomerular mesangial cells cultured in high glucose. J Am Soc Nephrol 14:873–880 - PubMed
-
- Anderson S, Jung FF, Ingelfinger JR 1993 Renal renin-angiotensin system in diabetes: functional, immunohistochemical, and molecular biological correlations. Am J Physiol 265:F477–F486 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

