The BC Cancer Agency Compassionate Access Program: outcome analysis of patients with esophagogastric cancer
- PMID: 19862357
- PMCID: PMC2768501
- DOI: 10.3747/co.v16i5.369
The BC Cancer Agency Compassionate Access Program: outcome analysis of patients with esophagogastric cancer
Abstract
Background: The BC Cancer Agency Gastro-intestinal Tumor Group supports one standard of care (SOC) chemotherapy regimen for metastatic esophagogastric adenocarcinoma-specifically, weekly cisplatin and 5-fluorouracil (5FU) infusion. All other regimens require Compassionate Access Program (CAP) approval for public funding.
Objectives: We examined response, toxicity, and survival after first-line cap chemotherapy (CAP1), or soc and second-line cap chemotherapy (CAP2).
Patients and methods: We searched cap records for December 1999 to April 2006, abstracted charts, constructed a database, and undertook survival analyses. Treatment response, serious toxicities, and hospitalizations were recorded.
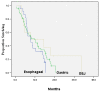
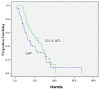
Results: We identified 32 esophageal (10 gastroesophageal junction) and 53 gastric cancer (62%) patients, 55 of whom were stage M1 at diagnosis. Prior therapy consisted of chemoradiotherapy (n = 14), adjuvant chemotherapy (n = 3), and radical surgery (n = 34). Of these 85 patients, 50 received CAP1, and 35 received soc, then CAP2. Docetaxel and irinotecan regimens accounted for 34% and 36%, 5% and 55%, 16% and 32% respectively of first-, second-, and third-line cap requests. Partial responses were documented with soc (11/35, 31%) and CAP1 (6/50, 12%). Grade 3+ toxicity rates were 19/50 (38%) and 6/35 (17%) with CAP1 and soc chemotherapy. With cap chemotherapy, 20 hospitalizations occurred, and with soc chemotherapy, 2 hospitalizations. For all patients, median follow-up and survival times were 8.9 months and 9.7 months respectively.
Limitations: This is a retrospective analysis of patients deemed suitable to receive non-soc chemotherapy regimens or unsuitable to receive soc chemotherapy.
Conclusions: Toxicities in cap chemotherapy regimens were substantial. Survival times were consistent with results of international phase II and III trials in esophagogastric cancer.
Keywords: Esophagogastric adenocarcinoma; chemotherapy; survival; toxicity.
Figures
Similar articles
-
Efficacy and safety of first-line systemic chemotherapy with epirubicin, cisplatin plus 5-fluorouracil and docetaxel, cisplatin plus 5-fluorouracil regimens in locally advanced inoperable or metastatic gastric or gastroesophageal junction adenocarcinoma: A prospective phase II study from South India.Indian J Cancer. 2017 Jan-Mar;54(1):47-51. doi: 10.4103/ijc.IJC_168_17. Indian J Cancer. 2017. PMID: 29199662 Clinical Trial.
-
Adjuvant (cisplatin, etoposide, and 5-fluorouracil) chemotherapy after curative resection of gastric adenocarcinomas involving the esophagogastric junction.Am J Clin Oncol. 1999 Jun;22(3):253-7. doi: 10.1097/00000421-199906000-00008. Am J Clin Oncol. 1999. PMID: 10362331 Clinical Trial.
-
Docetaxel, Oxaliplatin, and 5-Fluorouracil (DOF) in Metastatic and Unresectable Gastric/Gastroesophageal Junction Adenocarcinoma: A Phase II Study with Long-Term Follow-Up.Oncologist. 2019 Aug;24(8):1039-e642. doi: 10.1634/theoncologist.2019-0330. Epub 2019 May 28. Oncologist. 2019. PMID: 31138725 Free PMC article. Clinical Trial.
-
[Neoadjuvant chemoradiotherapy combined with surgery versus direct surgery in the treatment of Siewert type II and III adenocarcinomas of the esophagogastric junction: long-term prognostic analysis of a prospective randomized controlled trial].Zhonghua Wei Chang Wai Ke Za Zhi. 2021 Feb 25;24(2):128-137. doi: 10.3760/cma.j.cn.441530-20201019-00565. Zhonghua Wei Chang Wai Ke Za Zhi. 2021. PMID: 33508918 Clinical Trial. Chinese.
-
Expert opinion on management of gastric and gastro-oesophageal junction adenocarcinoma on behalf of the European Organisation for Research and Treatment of Cancer (EORTC)-gastrointestinal cancer group.Eur J Cancer. 2008 Jan;44(2):182-94. doi: 10.1016/j.ejca.2007.11.001. Eur J Cancer. 2008. PMID: 18093827
References
-
- Urba SG, Orringer MB, Turrisi A, Iannettoni M, Forastiere A, Strawderman M. Randomized trial of pre-operative chemoradiation versus surgery alone in patients with locoregional esophageal cancer. J Clin Oncol. 2000;19:303–13. - PubMed
-
- Herskovic A, Martz K, Al-Sarraf M, et al. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med. 1992;326:1593–8. - PubMed
-
- Macdonald JS, Smalley SR, Benedetti J, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001;345:725–30. - PubMed
-
- BC Cancer Agency (bcca) BCCA Protocol Summary for Combined Modality Therapy for Locally Advanced Esophageal Cancer using Cisplatin, Infusional Fluorouracil and Radiation Therapy. Vancouver: bcca; 1998. [Available online at: www.bccancer.bc.ca/NR/rdonlyres/C0A8933B-3AAD-4EBA-A95C-4552A0A060C9/339...; cited August 12, 2009]
-
- BC Cancer Agency (bcca) BCCA Protocol Summary for Combined Modality Adjuvant Therapy for Completely Resected Gastric Adenocarcinoma using Fluorouracil + Folinic Acid (Leucovorin) + Radiation Therapy. Vancouver: bcca; 2001. [Available online at: www.bccancer.bc.ca/NR/rdonlyres/C0A8933B-3AAD-4EBA-A95C-4552A0A060C9/339...; cited August 12, 2009]
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous