Long-term outcome of EBV-specific T-cell infusions to prevent or treat EBV-related lymphoproliferative disease in transplant recipients
- PMID: 19880495
- PMCID: PMC2817637
- DOI: 10.1182/blood-2009-08-239186
Long-term outcome of EBV-specific T-cell infusions to prevent or treat EBV-related lymphoproliferative disease in transplant recipients
Abstract
T-cell immunotherapy that takes advantage of Epstein-Barr virus (EBV)-stimulated immunity has the potential to fill an important niche in targeted therapy for EBV-related cancers. To address questions of long-term efficacy, safety, and practicality, we studied 114 patients who had received infusions of EBV-specific cytotoxic T lymphocytes (CTLs) at 3 different centers to prevent or treat EBV(+) lymphoproliferative disease (LPD) arising after hematopoietic stem cell transplantation. Toxicity was minimal, consisting mainly of localized swelling at sites of responsive disease. None of the 101 patients who received CTL prophylaxis developed EBV(+) LPD, whereas 11 of 13 patients treated with CTLs for biopsy-proven or probable LPD achieved sustained complete remissions. The gene-marking component of this study enabled us to demonstrate the persistence of functional CTLs for up to 9 years. A preliminary analysis indicated that a patient-specific CTL line can be manufactured, tested, and infused for $6095, a cost that compares favorably with other modalities used in the treatment of LPD. We conclude that the CTL lines described here provide safe and effective prophylaxis or treatment for lymphoproliferative disease in transplantation recipients, and the manufacturing methodology is robust and can be transferred readily from one institution to another without loss of reproducibility.
Trial registration: ClinicalTrials.gov NCT00058812.
Figures
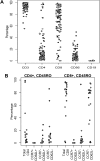

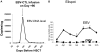

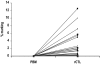

Comment in
-
EBV meets its match.Blood. 2010 Feb 4;115(5):920-1. doi: 10.1182/blood-2009-11-252437. Blood. 2010. PMID: 20133473 No abstract available.
References
-
- Reichert JM, Rosensweig CJ, Faden LB, Dewitz MC. Monoclonal antibody successes in the clinic. Nat Biotechnol. 2005;23(9):1073–1078. - PubMed
-
- Leen AM, Rooney CM, Foster AE. Improving T cell therapy for cancer. Annu Rev Immunol. 2007;25:243–265. - PubMed
-
- Blattman JN, Greenberg PD. Cancer immunotherapy: a treatment for the masses. Science. 2004;305(5681):200–205. - PubMed
-
- Appelbaum FR. Hematopoietic-cell transplantation at 50. N Engl J Med. 2007;357(15):1472–1475. - PubMed
-
- Bleakley M, Riddell SR. Molecules and mechanisms of the graft-versus-leukaemia effect. Nat Rev Cancer. 2004;4(5):371–380. - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

