The microcytic red cell and the anemia of inflammation
- PMID: 19890136
- PMCID: PMC3741048
- DOI: 10.1056/NEJMcibr0906391
The microcytic red cell and the anemia of inflammation
Conflict of interest statement
No potential conflict of interest relevant to this article was reported.
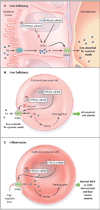
Figures
References
-
- Means R. Anemia of chronic disease. In: Young NS, Gerson SL, High KA, editors. Clinical hematology. Philadelphia: Mosby Elsevier; 2006.
-
- De Domenico I, McVey Ward D, Kaplan J. Regulation of iron acquisition and storage: consequences for iron-linked disorders. Nat Rev Mol Cell Biol. 2008;9:72–81. - PubMed
-
- Nemeth E, Tuttle MS, Powelson J, et al. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science. 2004;306:2090–2093. - PubMed
-
- Theurl I, Aigner E, Theurl M, et al. Regulation of iron homeostasis in anemia of chronic disease and iron deficiency anemia: diagnostic and therapeutic implications. Blood. 2009;113:5277–5286. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
