The acinar cell and early pancreatitis responses
- PMID: 19896090
- PMCID: PMC3073378
- DOI: 10.1016/j.cgh.2009.07.036
The acinar cell and early pancreatitis responses
Abstract
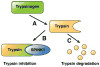
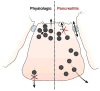
Pathologic responses arising from the pancreatic acinar cell appear to have a central role in initiating acute pancreatitis. Environmental factors that sensitize the acinar cell to harmful stimuli likely have a critical role in many forms of pancreatitis, including that induced by alcohol abuse. Activation of zymogens within the acinar cell and an inhibition of secretion are critical, but poorly understood, early pancreatitis events. While there is firm evidence relating trypsinogen activation to pancreatitis, the importance of other zymogens has been less studied. Preliminary studies suggest that trypsin may be activated by mechanisms that are distinct from other zymogens. Further, unlike the small intestine, it may not catalyze the activation of other zymogens. These features could affect strategies aimed at inhibiting proteases to treat pancreatitis. Specific intracellular signals are required to activate pancreatitis pathways in the acinar cell. The most important is calcium. Recent studies have suggested that calcium release through specific calcium channels in the endoplasmic reticulum is the means by which pathological elevations in cytosolic calcium occur. Although the targets of abnormal calcium signaling are unknown, calcineurin, a calcium-dependent phosphatase, may serve such a role. Finally, recent work suggests that an acute acid load might sensitize the acinar cell to pancreatitis responses. Therapies aimed at preventing or reversing the effects of an acid load on the pancreas may be important for treatment.
Conflict of interest statement
Conflict of interest
The authors disclose no conflicts.
Figures

References
-
- Murphy JA, Criddle DN, Sherwood M, et al. Direct activation of cytosolic Ca2+ signaling and enzyme secretion by cholecystokinin in human pancreatic acinar cells. Gastroenterology. 2008;135:632–641. - PubMed
-
- Katz M, Carangelo R, Miller LJ, et al. Effect of ethanol on chole-cystokinin-stimulated zymogen conversion in pancreatic acinar cells. Am J Physiol. 1996;270:G171–G175. - PubMed
-
- Pandol SJ, Periskic S, Gukovsky I, et al. Ethanol diet increases the sensitivity of rats to pancreatitis induced by cholecystokinin octapeptide. Gastroenterology. 1999;117:706–716. - PubMed
-
- Cosen-Binker LI, Lam PP, Binker MG, et al. Alcohol-induced protein kinase Calpha phosphorylation of Munc18c in carbachol-stimulated acini causes basolateral exocytosis. Gastroenterology. 2007;132:1527–1545. - PubMed
-
- Whitcomb DC, Gorry MC, Preston RA, et al. Hereditary pancreatitis is caused by a mutation on the cationic trypsinogen gene. Nat Genet. 1996;14:141–145. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

