Tetraspanin CD81 is required for Listeria monocytogenes invasion
- PMID: 19901060
- PMCID: PMC2798227
- DOI: 10.1128/IAI.00661-09
Tetraspanin CD81 is required for Listeria monocytogenes invasion
Abstract
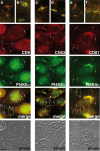
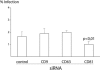
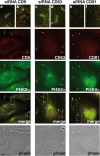
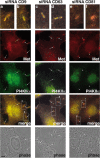
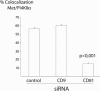
Listeria monocytogenes is an intracellular bacterial pathogen that invades epithelial cells by subverting two cellular receptors, E-cadherin and Met. We recently identified type II phosphatidylinositol 4-kinases alpha and beta (PI4KIIalpha and PI4KIIbeta) as being required for bacterial entry downstream of Met. In this work, we investigated whether tetraspanins CD9, CD63, and CD81, which figure among the few described molecular partners of PI4KIIalpha, function as molecular adaptors recruiting PI4KIIalpha to the bacterial entry site. We observed by fluorescence microscopy that CD9, CD63, and CD81 are expressed and detected at the cellular surface and also within intracellular compartments, particularly in the case of CD63. In resting cells, colocalization of tetraspanins and PI4KIIalpha is detectable only in restricted areas of the perinuclear region. Upon infection with Listeria, endogenous CD9, CD63, and CD81 were recruited to the bacterial entry site but did not colocalize strictly with endogenous PI4KIIalpha. Live-cell imaging confirmed that tetraspanins and PI4KIIalpha do not follow the same recruitment dynamics to the Listeria entry site. Depletion of CD9, CD63, and CD81 levels by small interfering RNA demonstrated that CD81 is required for bacterial internalization, identifying for the first time a role for a member of the tetraspanin family in the entry of Listeria into target cells. Moreover, depletion of CD81 inhibits the recruitment of PI4KIIalpha but not that of the Met receptor to the bacterial entry site, suggesting that CD81 may act as a membrane organizer required for the integrity of signaling events occurring at Listeria entry sites.
Figures


References
-
- Berditchevski, F., K. F. Tolias, K. Wong, C. L. Carpenter, and M. E. Hemler. 1997. A novel link between integrins, transmembrane-4 superfamily proteins (CD63 and CD81), and phosphatidylinositol 4-kinase. J. Biol. Chem. 272:2595-2598. - PubMed
-
- Braun, L., F. Nato, B. Payrastre, J. C. Mazie, and P. Cossart. 1999. The 213-amino-acid leucine-rich repeat region of the Listeria monocytogenes InlB protein is sufficient for entry into mammalian cells, stimulation of PI 3-kinase and membrane ruffling. Mol. Microbiol. 34:10-23. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

