Potent inhibition of HIV-1 replication by a Tat mutant
- PMID: 19901984
- PMCID: PMC2768900
- DOI: 10.1371/journal.pone.0007769
Potent inhibition of HIV-1 replication by a Tat mutant
Abstract
Herein we describe a mutant of the two-exon HIV-1 Tat protein, termed Nullbasic, that potently inhibits multiple steps of the HIV-1 replication cycle. Nullbasic was created by replacing the entire arginine-rich basic domain of wild type Tat with glycine/alanine residues. Like similarly mutated one-exon Tat mutants, Nullbasic exhibited transdominant negative effects on Tat-dependent transactivation. However, unlike previously reported mutants, we discovered that Nullbasic also strongly suppressed the expression of unspliced and singly-spliced viral mRNA, an activity likely caused by redistribution and thus functional inhibition of HIV-1 Rev. Furthermore, HIV-1 virion particles produced by cells expressing Nullbasic had severely reduced infectivity, a defect attributable to a reduced ability of the virions to undergo reverse transcription. Combination of these inhibitory effects on transactivation, Rev-dependent mRNA transport and reverse transcription meant that permissive cells constitutively expressing Nullbasic were highly resistant to a spreading infection by HIV-1. Nullbasic and its activities thus provide potential insights into the development of potent antiviral therapeutics that target multiple stages of HIV-1 infection.
Conflict of interest statement
Figures

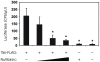
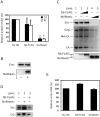

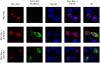
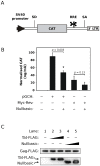
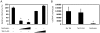
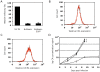
References
-
- Dayton AI, Sodroski JG, Rosen CA, Goh WC, Haseltine WA. The trans-activator gene of the human T cell lymphotropic virus type III is required for replication. Cell. 1986;44:941–947. - PubMed
-
- Berkhout B, Silverman RH, Jeang KT. Tat trans-activates the human immunodeficiency virus through a nascent RNA target. Cell. 1989;59:273–282. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

