Important precautions when deriving patient-specific neural elements from pluripotent cells
- PMID: 19903095
- PMCID: PMC3449142
- DOI: 10.3109/14653240903180092
Important precautions when deriving patient-specific neural elements from pluripotent cells
Abstract
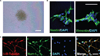
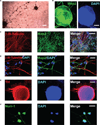
Multipotent human neural stem cells (hNSC) have traditionally been isolated directly from the central nervous system (CNS). To date, as a therapeutic tool in the treatment of neurologic disorders, the most promising results have been obtained using hNSC isolated directly from the human fetal neuroectoderm. The propagation ability of such tissue-derived hNSC is often limited, however, making it difficult to establish a large-scale culture. Following engraftment, these hNSC often show low efficiency in generating the desired neuronal cells necessary for reconstruction of the damaged host milieu and, as a result, have failed to give satisfactory results in clinical trials so far. Alternatively, human embryonic stem cells (hESC) offer a pluripotent reservoir for in vitro derivation of a rich spectrum of well-characterized neural-lineage committed stem/progenitor/precursor cells that can, theoretically, be picked at precisely their safest and most efficacious state of plasticity to meet a given clinical challenge. However, the need for 'foreign' biologic additives and multilineage differentiation inclination may make direct use of such cell-derived hNSC in patients problematic. The hNSC, when derived from pluripotent cells under protocols presently employed in the field, tend to display not only a low efficiency in neuronal differentiation, but also an inclination for phenotypic heterogeneity and instability and, hence, increased risk of tumorigenesis following engraftment. For hNSC derived in vitro to be used safely in therapeutic paradigms, it requires conversion of human pluripotent cells uniformly to cells that are restricted to the neural lineage in need of repair. Developing strategies for direct induction of human pluripotent cells exclusively into neural-committed progenies at a broad range of developmental stages will allow a large supply of optimal therapeutic hNSC tailor-made for safe and effective treatment of particular neurologic diseases and injuries in patients.
Conflict of interest statement
Figures
Similar articles
-
Efficient derivation of human neuronal progenitors and neurons from pluripotent human embryonic stem cells with small molecule induction.J Vis Exp. 2011 Oct 28;(56):e3273. doi: 10.3791/3273. J Vis Exp. 2011. PMID: 22064669 Free PMC article.
-
An Engraftable Human Embryonic Stem Cell Neuronal Lineage-Specific Derivative Retains Embryonic Chromatin Plasticity for Scale-Up CNS Regeneration.J Regen Med Tissue Eng. 2012 Sep 23;1(1):3. doi: 10.7243/2050-1218-1-3. J Regen Med Tissue Eng. 2012. PMID: 23542901 Free PMC article.
-
Efficient derivation of human cardiac precursors and cardiomyocytes from pluripotent human embryonic stem cells with small molecule induction.J Vis Exp. 2011 Nov 3;(57):e3274. doi: 10.3791/3274. J Vis Exp. 2011. PMID: 22083019 Free PMC article.
-
Differentiation of neural lineage cells from human pluripotent stem cells.Methods. 2008 Jun;45(2):142-58. doi: 10.1016/j.ymeth.2008.03.007. Epub 2008 May 29. Methods. 2008. PMID: 18593611 Free PMC article. Review.
-
A plethora of human pluripotent stem cells.Cell Biol Int. 2013 Sep;37(9):875-87. doi: 10.1002/cbin.10120. Epub 2013 May 23. Cell Biol Int. 2013. PMID: 23619972 Review.
Cited by
-
Current stem cell treatments for spinal cord injury.Indian J Orthop. 2012 Jan;46(1):10-8. doi: 10.4103/0019-5413.91629. Indian J Orthop. 2012. PMID: 22345801 Free PMC article. No abstract available.
-
Human Stem Cell Derivatives Retain More Open Epigenomic Landscape When Derived from Pluripotent Cells than from Tissues.J Regen Med. 2013 Jan 25;1(2):1000103. doi: 10.4172/2325-9620.1000103. J Regen Med. 2013. PMID: 23936871 Free PMC article.
-
Constraining the Pluripotent Fate of Human Embryonic Stem Cells for Tissue Engineering and Cell Therapy - The Turning Point of Cell-Based Regenerative Medicine.Br Biotechnol J. 2013 Oct 1;3(4):424-457. doi: 10.9734/BBJ/2013/4309#sthash.6D8Rulbv.dpuf. Br Biotechnol J. 2013. PMID: 24926434 Free PMC article.
-
Comparative study on the therapeutic potential of neurally differentiated stem cells in a mouse model of multiple sclerosis.PLoS One. 2012;7(4):e35093. doi: 10.1371/journal.pone.0035093. Epub 2012 Apr 13. PLoS One. 2012. PMID: 22514711 Free PMC article.
-
MicroRNA Profiling Reveals Distinct Mechanisms Governing Cardiac and Neural Lineage-Specification of Pluripotent Human Embryonic Stem Cells.J Stem Cell Res Ther. 2012 Jul 13;2(3):124. doi: 10.4172/2157-7633.1000124. J Stem Cell Res Ther. 2012. PMID: 23355957 Free PMC article.
References
-
- Park KI, Ourednik J, Ourednik V, Taylor RM, Aboody KA, Auguste KI, et al. Global gene and cell replacement strategies via stem cells. Gene Therapy. 2002;9:613–624. - PubMed
-
- Park KI, Teng YD, Snyder EY. The injured brain interacts reciprocally with neural stem cells supported by scaffolds to reconstitute lost tissue. Nat Biotechnol. 2002;20:1111–1117. - PubMed
-
- Ourednik J, Ourednik V, Lynch WP, Schachner M, Snyder EY. Neural stem cells display an inherent mechanism for rescuing dysfunctional neurons. Nat Biotechnol. 2002;20:1103–1110. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

