Vitamin B6 deficient plants display increased sensitivity to high light and photo-oxidative stress
- PMID: 19903353
- PMCID: PMC2777905
- DOI: 10.1186/1471-2229-9-130
Vitamin B6 deficient plants display increased sensitivity to high light and photo-oxidative stress
Abstract
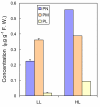
Background: Vitamin B6 is a collective term for a group of six interconvertible compounds: pyridoxine, pyridoxal, pyridoxamine and their phosphorylated derivatives. Vitamin B6 plays essential roles as a cofactor in a range of biochemical reactions. In addition, vitamin B6 is able to quench reactive oxygen species in vitro, and exogenously applied vitamin B6 protects plant cells against cell death induced by singlet oxygen (1O2). These results raise the important question as to whether plants employ vitamin B6 as an antioxidant to protect themselves against reactive oxygen species.
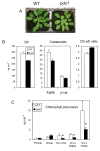
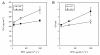
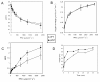
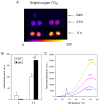
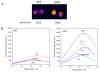
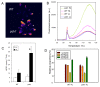
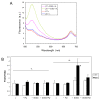
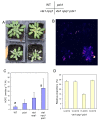
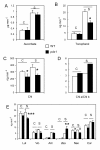
Results: The pdx1.3 mutation affects the vitamin B6 biosynthesis enzyme, pyridoxal synthase (PDX1), and leads to a reduction of the vitamin B6 concentration in Arabidopsis thaliana leaves. Although leaves of the pdx1.3 Arabidopsis mutant contained less chlorophyll than wild-type leaves, we found that vitamin B6 deficiency did not significantly impact photosynthetic performance or shoot and root growth. Chlorophyll loss was associated with an increase in the chlorophyll a/b ratio and a selective decrease in the abundance of several PSII antenna proteins (Lhcb1/2, Lhcb6). These changes were strongly dependent on light intensity, with high light amplifying the difference between pdx1.3 and the wild type. When leaf discs were exposed to exogenous 1O2, lipid peroxidation in pdx1.3 was increased relative to the wild type; this effect was not observed with superoxide or hydrogen peroxide. When leaf discs or whole plants were exposed to excess light energy, 1O2-mediated lipid peroxidation was enhanced in leaves of the pdx1.3 mutant relative to the wild type. High light also caused an increased level of 1O2 in vitamin B6-deficient leaves. Combining the pdx1.3 mutation with mutations affecting the level of 'classical' quenchers of 1O2 (zeaxanthin, tocopherols) resulted in a highly photosensitive phenotype.
Conclusion: This study demonstrates that vitamin B6 has a function in the in vivo antioxidant defense of plants. Thus, the antioxidant activity of vitamin B6 inferred from in vitro studies is confirmed in planta. Together with the finding that chloroplasts contain vitamin B6 compounds, the data show that vitamin B6 functions as a photoprotector that limits 1O2 accumulation in high light and prevents 1O2-mediated oxidative damage.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

