Regulation of steroidogenic acute regulatory protein transcription in largemouth bass by orphan nuclear receptor signaling pathways
- PMID: 19906818
- PMCID: PMC2803149
- DOI: 10.1210/en.2009-0551
Regulation of steroidogenic acute regulatory protein transcription in largemouth bass by orphan nuclear receptor signaling pathways
Abstract
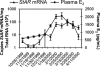
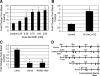
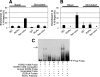
The steroidogenic acute regulatory (StAR) protein mediates the rate-limiting step of mitochondrial transport of cholesterol for steroid biosynthesis. To investigate the regulation of this protein in lower vertebrates, we cloned the StAR coding region from large-mouth bass for analysis. Induction of the mRNA corresponded with increasing levels of plasma sex steroids in vivo. Cultures of largemouth bass ovarian follicles were exposed to dibutyryl cAMP (dbcAMP), a potent signaling molecule for steroidogenesis. StAR mRNA expression was significantly up-regulated by dbcAMP signaling, suggesting that the 5' regulatory region of the gene is functionally conserved. To further analyze its transcriptional regulation, a 2.9-kb portion of the promoter was cloned and transfected into Y-1 cells, a steroidogenic mouse adrenocortical cell line. The promoter activity was induced in a dose-responsive manner upon stimulation with dbcAMP; however, deletion of 1 kb from the 5' end of the promoter segment significantly diminished the transcriptional activation. A putative retinoic acid-related receptor-alpha/rev-erb alpha element was identified between the -1.86- and -2.9-kb region and mutated to assess its potential role in dbcAMP regulation of the promoter. Mutation of the rev-erb alpha element significantly impeded dbcAMP-induced activation. Chromatin immunoprecipitation and EMSA results revealed rev-erb alpha and retinoic acid-related receptor-alpha enrichment at the site under basal and dbcAMP-induced conditions, respectively. These results implicate important roles for these proteins previously uncharacterized for the StAR promoter. Altogether these data suggest novel regulatory mechanisms for dbcAMP up-regulation of StAR transcription in the distal part of the largemouth bass promoter.
Figures

Similar articles
-
Steroidogenic acute regulatory protein transcription is regulated by estrogen receptor signaling in largemouth bass ovary.Gen Comp Endocrinol. 2020 Jan 15;286:113300. doi: 10.1016/j.ygcen.2019.113300. Epub 2019 Oct 31. Gen Comp Endocrinol. 2020. PMID: 31678557 Free PMC article.
-
Regulation of steroidogenesis and the steroidogenic acute regulatory protein by a member of the cAMP response-element binding protein family.Mol Endocrinol. 2002 Jan;16(1):184-99. doi: 10.1210/mend.16.1.0759. Mol Endocrinol. 2002. PMID: 11773448
-
Characterization of the promoter region of the mouse gene encoding the steroidogenic acute regulatory protein.Mol Endocrinol. 1997 Feb;11(2):138-47. doi: 10.1210/mend.11.2.9880. Mol Endocrinol. 1997. PMID: 9013761
-
Regulation of expression of the steroidogenic acute regulatory protein (StAR) gene: a central role for steroidogenic factor 1.Steroids. 1997 Jan;62(1):5-9. doi: 10.1016/s0039-128x(96)00152-3. Steroids. 1997. PMID: 9029708 Review.
-
Involvement of multiple transcription factors in the regulation of steroidogenic acute regulatory protein gene expression.Steroids. 2003 Dec;68(14):1125-34. doi: 10.1016/j.steroids.2003.07.009. Steroids. 2003. PMID: 14643873 Review.
Cited by
-
Steroidogenic acute regulatory protein transcription is regulated by estrogen receptor signaling in largemouth bass ovary.Gen Comp Endocrinol. 2020 Jan 15;286:113300. doi: 10.1016/j.ygcen.2019.113300. Epub 2019 Oct 31. Gen Comp Endocrinol. 2020. PMID: 31678557 Free PMC article.
-
Cloning and expression of the translocator protein (18 kDa), voltage-dependent anion channel, and diazepam binding inhibitor in the gonad of largemouth bass (Micropterus salmoides) across the reproductive cycle.Gen Comp Endocrinol. 2011 Aug 1;173(1):86-95. doi: 10.1016/j.ygcen.2011.04.027. Epub 2011 May 10. Gen Comp Endocrinol. 2011. PMID: 21600210 Free PMC article.
-
Carotenoid coloration and coloration-linked gene expression in red tilapia (Oreochromis sp.) tissues.BMC Vet Res. 2021 Sep 25;17(1):314. doi: 10.1186/s12917-021-03006-5. BMC Vet Res. 2021. PMID: 34563199 Free PMC article.
-
Parabens Accelerate Ovarian Dysfunction in a 4-Vinylcyclohexene Diepoxide-Induced Ovarian Failure Model.Int J Environ Res Public Health. 2017 Feb 8;14(2):161. doi: 10.3390/ijerph14020161. Int J Environ Res Public Health. 2017. PMID: 28208728 Free PMC article.
-
Expression and Transcript Localization of star, sf-1, and dax-1 in the Early Brain of the Orange-Spotted Grouper Epinephelus coioides.Int J Mol Sci. 2022 Feb 27;23(5):2614. doi: 10.3390/ijms23052614. Int J Mol Sci. 2022. PMID: 35269757 Free PMC article.
References
-
- Clark BJ, Wells J, King SR, Stocco DM 1994 The purification, cloning, and expression of a novel luteinizing hormone-induced mitochondrial protein in MA-10 mouse Leydig tumor cells. Characterization of the steroidogenic acute regulatory protein (StAR). J Biol Chem 269:28314–28322 - PubMed
-
- Manna PR, Stocco DM 2005 Regulation of the steroidogenic acute regulatory protein expression: functional and physiological consequences. Curr Drug Targets Immune Endocr Metab Disord 5:93–108 - PubMed
-
- Clark BJ, Stocco DM 1995 Expression of the steroidogenic acute regulatory (StAR) protein: a novel LH-induced mitochondrial protein required for the acute regulation of steroidogenesis in mouse Leydig tumor cells. Endocr Res 21:243–257 - PubMed
-
- Stocco DM, Clark BJ 1996 Role of the steroidogenic acute regulatory protein (StAR) in steroidogenesis. Biochem Pharmacol 51:197–205 - PubMed
-
- Stocco DM, Clark BJ 1997 The role of the steroidogenic acute regulatory protein in steroidogenesis. Steroids 62:29–36 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

