Local and large-range inhibition in feature detection
- PMID: 19906963
- PMCID: PMC6665075
- DOI: 10.1523/JNEUROSCI.2857-09.2009
Local and large-range inhibition in feature detection
Abstract
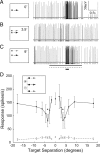
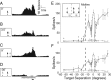
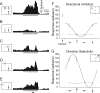
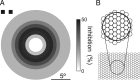
Lateral inhibition is perhaps the most ubiquitous of neuronal mechanisms, having been demonstrated in early stages of processing in many different sensory pathways of both mammals and invertebrates. Recent work challenges the long-standing view that assumes that similar mechanisms operate to tune neuronal responses to higher order properties. Scant evidence for lateral inhibition exists beyond the level of the most peripheral stages of visual processing, leading to suggestions that many features of the tuning of higher order visual neurons can be accounted for by the receptive field and other intrinsic coding properties of visual neurons. Using insect target neurons as a model, we present unequivocal evidence that feature tuning is shaped not by intrinsic properties but by potent spatial lateral inhibition operating well beyond the first stages of visual processing. In addition, we present evidence for a second form of higher-order spatial inhibition--a long-range interocular transfer of information that we argue serves a role in establishing interocular rivalry and thus potentially a neural substrate for directing attention to single targets in the presence of distracters. In so doing, we demonstrate not just one, but two levels of spatial inhibition acting beyond the level of peripheral processing.
Figures



References
-
- Baird JM, May ML. Foraging behavior of Pachydiplax longipennis (Odonata:Libellulidae) J Insect Behav. 1997;10:655–678.
-
- Baird JM, May ML. Fights at the dinner table: agonistic behavior in Pachydiplax longipennis (Odonata:Libellulidae) at feeding sites. J Insect Behav. 2003;16:189–216.
-
- Barnett PD, Nordström K, O'Carroll DC. Retinotopic organisation of small-field-target-detecting neurons in the insect visual system. Curr Biol. 2007;17:569–578. - PubMed
-
- Braitenberg V. Periodic structures in the visual system of the fly. In: Wehner R, editor. Information processing in the visual systems of arthropods. Berlin, Heidelberg: Springer; 1972. pp. 3–15.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
