The role of oestradiol in sexually dimorphic hypothalamic-pituitary-adrena axis responses to intracerebroventricular ethanol administration in the rat
- PMID: 19912475
- PMCID: PMC2818645
- DOI: 10.1111/j.1365-2826.2009.01934.x
The role of oestradiol in sexually dimorphic hypothalamic-pituitary-adrena axis responses to intracerebroventricular ethanol administration in the rat
Abstract
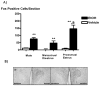
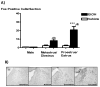
Systemic ethanol (EtOH) administration activates the hypothalamic-pituitary-adrenal (HPA) axis of rats in a sexually dimorphic manner. The present studies tested the role played by the central nervous system (CNS) in this phenomenon. To localise the effects of the drug to the brain, we utilised an experimental paradigm whereby a small, nontoxic amount of the drug was delivered via intracerebroventricular (i.c.v.) injection. EtoH administered i.c.v. rapidly diffuses throughout the cerebrospinal fluid and brain, and does not cause neuronal damage or have any long-term physiological or behavioural effects. Experimental groups included intact males, intact cycling females, and ovariectomised (OVX) animals with or without replacement oestradiol (E(2)). Intracerebroventricular EtOH-induced HPA hormonal activation was determined by measuring plasma adrenocorticotrophin (ACTH) levels. Activation of brain areas that both regulate HPA function and are responsive to gonadal hormones was determined using expression of the transcription factor c-fos (Fos) as a marker of neuronal activity. We observed sex- and oestrous cycle- dependent differences in HPA activation by EtOH as measured by both these parameters. ACTH secretion was highest in females in pro-oestrus or oestrus, just prior to or after the endogenous peak of E(2), as was Fos expression in the paraventricular nucleus of the hypothalamus (PVN) and the locus coreuleus (LC) of the brainstem. In OVX animals, E(2) replacement caused an increase in PVN and LC Fos expression in response to i.c.v. EtOH compared to OVX controls, but a decrease in ACTH secretion. Taken together, these results indicate that at the level of the CNS, EtOH stimulates HPA activity more robustly at times when the effects of E(2) are high, but that E(2) alone is not responsible for this effect. The data further suggest that the LC plays an important role in the circuitry, which appears to be different from that activated following the systemic administration of EtOH.
Figures

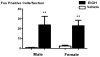
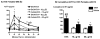
Similar articles
-
Roles of the locus coeruleus and adrenergic receptors in brain-mediated hypothalamic-pituitary-adrenal axis responses to intracerebroventricular alcohol.Alcohol Clin Exp Res. 2012 Jun;36(6):1084-90. doi: 10.1111/j.1530-0277.2011.01707.x. Epub 2012 Jan 11. Alcohol Clin Exp Res. 2012. PMID: 22236039 Free PMC article.
-
Gender difference in hypothalamic-pituitary-adrenal axis response to alcohol in the rat: activational role of gonadal steroids.Brain Res. 1997 Aug 22;766(1-2):19-28. doi: 10.1016/s0006-8993(97)00525-8. Brain Res. 1997. PMID: 9359583
-
Estrogen impairs glucocorticoid dependent negative feedback on the hypothalamic-pituitary-adrenal axis via estrogen receptor alpha within the hypothalamus.Neuroscience. 2009 Mar 17;159(2):883-95. doi: 10.1016/j.neuroscience.2008.12.058. Epub 2009 Jan 7. Neuroscience. 2009. PMID: 19166915 Free PMC article.
-
Mechanisms mediating the influence of alcohol on the hypothalamic-pituitary-adrenal axis responses to immune and nonimmune signals.Alcohol Clin Exp Res. 1998 Aug;22(5 Suppl):243S-247S. doi: 10.1097/00000374-199805001-00005. Alcohol Clin Exp Res. 1998. PMID: 9727644 Review.
-
Role of various neurotransmitters in mediating the long-term endocrine consequences of prenatal alcohol exposure.Ann N Y Acad Sci. 2008 Nov;1144:176-88. doi: 10.1196/annals.1418.015. Ann N Y Acad Sci. 2008. PMID: 19076376 Free PMC article. Review.
Cited by
-
Sex differences in the hypothalamic-pituitary-adrenal axis' response to stress: an important role for gonadal hormones.Neuropsychopharmacology. 2019 Jan;44(1):45-58. doi: 10.1038/s41386-018-0167-9. Epub 2018 Aug 1. Neuropsychopharmacology. 2019. PMID: 30111811 Free PMC article. Review.
-
Neuroendocrine pathogenesis of perimenopausal depression.Front Psychiatry. 2023 Mar 30;14:1162501. doi: 10.3389/fpsyt.2023.1162501. eCollection 2023. Front Psychiatry. 2023. PMID: 37065890 Free PMC article. Review.
-
Central 5-alpha reduction of testosterone is required for testosterone's inhibition of the hypothalamo-pituitary-adrenal axis response to restraint stress in adult male rats.Brain Res. 2013 Sep 5;1529:74-82. doi: 10.1016/j.brainres.2013.07.021. Epub 2013 Jul 21. Brain Res. 2013. PMID: 23880372 Free PMC article.
-
Natural and Drug Rewards Engage Distinct Pathways that Converge on Coordinated Hypothalamic and Reward Circuits.Neuron. 2019 Sep 4;103(5):891-908.e6. doi: 10.1016/j.neuron.2019.05.050. Epub 2019 Jul 2. Neuron. 2019. PMID: 31277924 Free PMC article.
-
Sex Differences in Acute Neuroendocrine Responses to Stressors in Rodents and Humans.Cold Spring Harb Perspect Biol. 2022 Jun 6;14(9):a039081. doi: 10.1101/cshperspect.a039081. Online ahead of print. Cold Spring Harb Perspect Biol. 2022. PMID: 35667789 Free PMC article.
References
-
- Iwasaki-Sekino A, et al. Gender differences in corticotropin and corticosterone secretion and corticotropin-releasing factor mRNA expression in the paraventricular nucleus of the hypothalamus and the central nucleus of the amygdala in response to footshock stress or psychological stress in rats. Psychoneuroendocrinology. 2009;34(2):226–37. - PubMed
-
- Jezova D, et al. Neuroendocrine response during stress with relation to gender differences. Acta Neurobiol Exp (Wars) 1996;56(3):779–85. - PubMed
-
- Clarke TK, Schumann G. Gene-environment interactions resulting in risk alcohol drinking behaviour are mediated by CRF and CRF1. Pharmacol Biochem Behav. 2009;93(3):230–6. - PubMed
-
- Zilberman ML, et al. Substance use disorders: sex differences and psychiatric comorbidities. Can J Psychiatry. 2003;48(1):5–13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

