RNA adenosine deaminase ADAR1 deficiency leads to increased activation of protein kinase PKR and reduced vesicular stomatitis virus growth following interferon treatment
- PMID: 19913273
- PMCID: PMC2789878
- DOI: 10.1016/j.virol.2009.10.026
RNA adenosine deaminase ADAR1 deficiency leads to increased activation of protein kinase PKR and reduced vesicular stomatitis virus growth following interferon treatment
Abstract
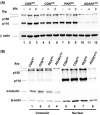
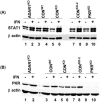
Two size forms of ADAR1 adenosine deaminase are known, one constitutively expressed (p110) and the other interferon (IFN)-induced (p150). To test the role of ADAR1 in viral infection, HeLa cells with ADAR1 stably knocked down and 293 cells overexpressing ADAR1 were utilized. Overexpression of p150 ADAR1 had no significant effect on the yield of vesicular stomatitis virus. Likewise, reduction of p110 and p150 ADAR1 proteins to less than approximately 10 to 15% of parental levels (ADAR1-deficient) had no significant effect on VSV growth in the absence of IFN treatment. However, inhibition of virus growth following IFN treatment was approximately 1 log(10) further reduced compared to ADAR1-sufficient cells. The level of phosphorylated protein kinase PKR was increased in ADAR1-deficient cells compared to ADAR1-sufficient cells following IFN treatment, regardless of viral infection. These results suggest that ADAR1 suppresses activation of PKR and inhibition of VSV growth in response to IFN treatment.
Figures



Similar articles
-
Adenosine deaminase acting on RNA 1 (ADAR1) suppresses the induction of interferon by measles virus.J Virol. 2012 Apr;86(7):3787-94. doi: 10.1128/JVI.06307-11. Epub 2012 Jan 25. J Virol. 2012. PMID: 22278222 Free PMC article.
-
Stress granule formation induced by measles virus is protein kinase PKR dependent and impaired by RNA adenosine deaminase ADAR1.J Virol. 2013 Jan;87(2):756-66. doi: 10.1128/JVI.02270-12. Epub 2012 Oct 31. J Virol. 2013. PMID: 23115276 Free PMC article.
-
RNA-specific adenosine deaminase ADAR1 suppresses measles virus-induced apoptosis and activation of protein kinase PKR.J Biol Chem. 2009 Oct 23;284(43):29350-6. doi: 10.1074/jbc.M109.045146. Epub 2009 Aug 25. J Biol Chem. 2009. PMID: 19710021 Free PMC article.
-
Protein kinase PKR and RNA adenosine deaminase ADAR1: new roles for old players as modulators of the interferon response.Curr Opin Immunol. 2011 Oct;23(5):573-82. doi: 10.1016/j.coi.2011.08.009. Epub 2011 Sep 15. Curr Opin Immunol. 2011. PMID: 21924887 Free PMC article. Review.
-
Tipping the balance: antagonism of PKR kinase and ADAR1 deaminase functions by virus gene products.J Interferon Cytokine Res. 2009 Sep;29(9):477-87. doi: 10.1089/jir.2009.0065. J Interferon Cytokine Res. 2009. PMID: 19715457 Free PMC article. Review.
Cited by
-
Type I interferon modulates the battle of host immune system against viruses.Adv Appl Microbiol. 2010;73:83-101. doi: 10.1016/S0065-2164(10)73004-5. Adv Appl Microbiol. 2010. PMID: 20800760 Free PMC article. Review.
-
Induction of stress granules by interferon and down-regulation by the cellular RNA adenosine deaminase ADAR1.Virology. 2014 Apr;454-455:299-310. doi: 10.1016/j.virol.2014.02.025. Epub 2014 Mar 21. Virology. 2014. PMID: 24725957 Free PMC article.
-
Adenosine deaminases acting on RNA (ADARs) are both antiviral and proviral.Virology. 2011 Mar 15;411(2):180-93. doi: 10.1016/j.virol.2010.12.004. Epub 2011 Jan 5. Virology. 2011. PMID: 21211811 Free PMC article. Review.
-
dsRNA-dependent protein kinase PKR and its role in stress, signaling and HCV infection.Viruses. 2012 Oct 29;4(11):2598-635. doi: 10.3390/v4112598. Viruses. 2012. PMID: 23202496 Free PMC article. Review.
-
ADAR1 restricts ZBP1-mediated immune response and PANoptosis to promote tumorigenesis.Cell Rep. 2021 Oct 19;37(3):109858. doi: 10.1016/j.celrep.2021.109858. Cell Rep. 2021. PMID: 34686350 Free PMC article.
References
-
- Athanasiadis A, Placido D, Maas S, Brown BA, Lowenhaupt K, Rich A. The crystal structure of the Z beta domain of the RNA-editing enzyme ADAR1 reveals distinct conserved surfaces among Z-domains. J. Mol. Biol. 2005;351:496–507. - PubMed
-
- Balachandran S, Roberts PC, Brown LE, Truong H, Pattnaik AK, Archer DR, Barber GN. Essential role for the dsRNA-dependent protein kinase PKR in innate immunity to viral infection. Immunity. 2000;13:129–141. - PubMed
-
- Bass BL, Weintraub H. An unwinding activity that covalently modifies its double-stranded RNA substrate. Cell. 1988;55:1089–1098. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

