Optically resolving individual microtubules in live axons
- PMID: 19913478
- PMCID: PMC2927971
- DOI: 10.1016/j.str.2009.09.008
Optically resolving individual microtubules in live axons
Abstract
Microtubules are essential cytoskeletal tracks for cargo transportation in axons and also serve as the primary structural scaffold of neurons. Structural assembly, stability, and dynamics of axonal microtubules are of great interest for understanding neuronal functions and pathologies. However, microtubules are so densely packed in axons that their separations are well below the diffraction limit of light, which precludes using optical microscopy for live-cell studies. Here, we present a single-molecule imaging method capable of resolving individual microtubules in live axons. In our method, unlabeled microtubules are revealed by following individual axonal cargos that travel along them. We resolved more than six microtubules in a 1 microm diameter axon by real-time tracking of endosomes containing quantum dots. Our live-cell study also provided direct evidence that endosomes switch between microtubules while traveling along axons, which has been proposed to be the primary means for axonal cargos to effectively navigate through the crowded axoplasmic environment.
Figures
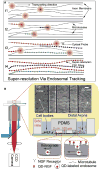
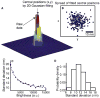
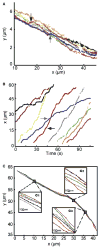
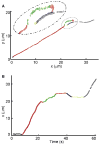
Similar articles
-
Live-cell imaging of neurofilament transport in cultured neurons.Methods Cell Biol. 2016;131:21-90. doi: 10.1016/bs.mcb.2015.07.001. Epub 2015 Sep 2. Methods Cell Biol. 2016. PMID: 26794508 Free PMC article.
-
Unrestrained growth of correctly oriented microtubules instructs axonal microtubule orientation.Elife. 2022 Oct 10;11:e77608. doi: 10.7554/eLife.77608. Elife. 2022. PMID: 36214669 Free PMC article.
-
Motor transport of self-assembled cargos in crowded environments.Proc Natl Acad Sci U S A. 2012 Dec 18;109(51):20814-9. doi: 10.1073/pnas.1209304109. Epub 2012 Dec 3. Proc Natl Acad Sci U S A. 2012. PMID: 23213204 Free PMC article.
-
The neuronal centrosome as a generator of microtubules for the axon.Curr Top Dev Biol. 1996;33:281-98. doi: 10.1016/s0070-2153(08)60341-5. Curr Top Dev Biol. 1996. PMID: 9138908 Review. No abstract available.
-
Microtubule transport in the axon.Int Rev Cytol. 2002;212:41-62. doi: 10.1016/s0074-7696(01)12003-6. Int Rev Cytol. 2002. PMID: 11804039 Review.
Cited by
-
HDAC6 mutations rescue human tau-induced microtubule defects in Drosophila.Proc Natl Acad Sci U S A. 2013 Mar 19;110(12):4604-9. doi: 10.1073/pnas.1207586110. Epub 2013 Mar 4. Proc Natl Acad Sci U S A. 2013. PMID: 23487739 Free PMC article.
-
Competitive dynamics during resource-driven neurite outgrowth.PLoS One. 2014 Feb 3;9(2):e86741. doi: 10.1371/journal.pone.0086741. eCollection 2014. PLoS One. 2014. PMID: 24498280 Free PMC article.
-
Intracellular transport dynamics revealed by single-particle tracking.Biophys Rep. 2021 Oct 31;7(5):413-427. doi: 10.52601/bpr.2021.210035. Biophys Rep. 2021. PMID: 37288105 Free PMC article.
-
The nano-architecture of the axonal cytoskeleton.Nat Rev Neurosci. 2017 Dec;18(12):713-726. doi: 10.1038/nrn.2017.129. Epub 2017 Nov 3. Nat Rev Neurosci. 2017. PMID: 29097785 Review.
-
Munc13-4 interacts with syntaxin 7 and regulates late endosomal maturation, endosomal signaling, and TLR9-initiated cellular responses.Mol Biol Cell. 2016 Feb 1;27(3):572-87. doi: 10.1091/mbc.E15-05-0283. Epub 2015 Dec 17. Mol Biol Cell. 2016. PMID: 26680738 Free PMC article.
References
-
- Andrieux A, Salin P, Schweitzer A, Begou M, Pachoud B, Brun P, Gory-Faure S, Kujala P, Suaud-Chagny MF, Hofle G, Job D. Microtubule stabilizer ameliorates synaptic function and behavior in a mouse model for schizophrenia. Biol Psychiatry. 2006;60:1224–1230. - PubMed
-
- Baas PW, Qiang L. Neuronal microtubules: when the MAP is the roadblock. Trends Cell Biol. 2005;15:183–187. - PubMed
-
- Betzig E, Patterson GH, Sougrat R, Lindwasser OW, Olenych S, Bonifacino JS, Davidson MW, Lippincott-Schwartz J, Hess HF. Imaging intracellular fluorescent proteins at nanometer resolution. Science. 2006;313:1642–1645. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

