Activation of aldehyde dehydrogenase 2 (ALDH2) confers cardioprotection in protein kinase C epsilon (PKCvarepsilon) knockout mice
- PMID: 19913552
- PMCID: PMC2837767
- DOI: 10.1016/j.yjmcc.2009.10.030
Activation of aldehyde dehydrogenase 2 (ALDH2) confers cardioprotection in protein kinase C epsilon (PKCvarepsilon) knockout mice
Abstract
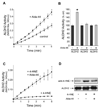
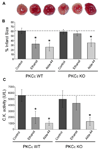
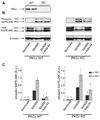
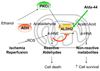
Acute administration of ethanol can reduce cardiac ischemia/reperfusion injury. Previous studies demonstrated that the acute cytoprotective effect of ethanol on the myocardium is mediated by protein kinase C epsilon (PKCvarepsilon). We recently identified aldehyde dehydrogenase 2 (ALDH2) as a PKCvarepsilon substrate, whose activation is necessary and sufficient to confer cardioprotection in vivo. ALDH2 metabolizes cytotoxic reactive aldehydes, such as 4-hydroxy-2-nonenal (4-HNE), which accumulate during cardiac ischemia/reperfusion. Here, we used a combination of PKCvarepsilon knockout mice and a direct activator of ALDH2, Alda-44, to further investigate the interplay between PKCvarepsilon and ALDH2 in cardioprotection. We report that ethanol preconditioning requires PKCvarepsilon, whereas direct activation of ALDH2 reduces infarct size in both wild type and PKCvarepsilon knockout hearts. Our data suggest that ALDH2 is downstream of PKCvarepsilon in ethanol preconditioning and that direct activation of ALDH2 can circumvent the requirement of PKCvarepsilon to induce cytoprotection. We also report that in addition to ALDH2 activation, Alda-44 prevents 4-HNE induced inactivation of ALDH2 by reducing the formation of 4-HNE-ALDH2 protein adducts. Thus, Alda-44 promotes metabolism of cytotoxic reactive aldehydes that accumulate in ischemic myocardium. Taken together, our findings suggest that direct activation of ALDH2 may represent a method of harnessing the cardioprotective effect of ethanol without the side effects associated with alcohol consumption.
Copyright (c) 2009 Elsevier Ltd. All rights reserved.
Figures



Similar articles
-
Time-dependent and ethanol-induced cardiac protection from ischemia mediated by mitochondrial translocation of varepsilonPKC and activation of aldehyde dehydrogenase 2.J Mol Cell Cardiol. 2009 Feb;46(2):278-84. doi: 10.1016/j.yjmcc.2008.09.713. Epub 2008 Oct 17. J Mol Cell Cardiol. 2009. PMID: 18983847 Free PMC article.
-
Aldehyde dehydrogenase 2 in cardiac protection: a new therapeutic target?Trends Cardiovasc Med. 2009 Jul;19(5):158-64. doi: 10.1016/j.tcm.2009.09.003. Trends Cardiovasc Med. 2009. PMID: 20005475 Free PMC article. Review.
-
Isoflurane preconditioning confers cardioprotection by activation of ALDH2.PLoS One. 2013;8(2):e52469. doi: 10.1371/journal.pone.0052469. Epub 2013 Feb 28. PLoS One. 2013. Retraction in: PLoS One. 2023 Sep 28;18(9):e0292458. doi: 10.1371/journal.pone.0292458. PMID: 23468836 Free PMC article. Retracted.
-
Aldehyde dehydrogenase 2 ameliorates doxorubicin-induced myocardial dysfunction through detoxification of 4-HNE and suppression of autophagy.J Mol Cell Cardiol. 2014 Jun;71:92-104. doi: 10.1016/j.yjmcc.2014.01.002. Epub 2014 Jan 13. J Mol Cell Cardiol. 2014. PMID: 24434637
-
Salvaging the Ischemic Heart: Gi-Coupled Receptors in Mast Cells Activate a PKCε/ALDH2 Pathway Providing Anti-RAS Cardioprotection.Curr Med Chem. 2018;25(34):4416-4431. doi: 10.2174/0929867325666180214115127. Curr Med Chem. 2018. PMID: 29446730 Review.
Cited by
-
Cardioprotection induced by a brief exposure to acetaldehyde: role of aldehyde dehydrogenase 2.Cardiovasc Res. 2018 Jun 1;114(7):1006-1015. doi: 10.1093/cvr/cvy070. Cardiovasc Res. 2018. PMID: 29579152 Free PMC article.
-
Moderate ethanol ingestion and cardiovascular protection: from epidemiologic associations to cellular mechanisms.J Mol Cell Cardiol. 2012 Jan;52(1):93-104. doi: 10.1016/j.yjmcc.2011.10.011. Epub 2011 Oct 23. J Mol Cell Cardiol. 2012. PMID: 22041278 Free PMC article. Review.
-
Development of selective inhibitors for human aldehyde dehydrogenase 3A1 (ALDH3A1) for the enhancement of cyclophosphamide cytotoxicity.Chembiochem. 2014 Mar 21;15(5):701-12. doi: 10.1002/cbic.201300625. Chembiochem. 2014. PMID: 24677340 Free PMC article.
-
Mitochondrial aldehyde dehydrogenase-2 deficiency compromises therapeutic effect of ALDH bright cell on peripheral ischemia.Redox Biol. 2017 Oct;13:196-206. doi: 10.1016/j.redox.2017.05.018. Epub 2017 May 29. Redox Biol. 2017. PMID: 28582728 Free PMC article.
-
Role of Alcohol Oxidative Metabolism in Its Cardiovascular and Autonomic Effects.Adv Exp Med Biol. 2019;1193:1-33. doi: 10.1007/978-981-13-6260-6_1. Adv Exp Med Biol. 2019. PMID: 31368095 Free PMC article. Review.
References
-
- Kloner RA, Rezkalla SH. To drink or not to drink? That is the question. Circulation. 2007;116(11):1306–1317. - PubMed
-
- Renaud S, de Lorgeril M. Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet. 1992;339(8808):1523–1526. - PubMed
-
- Chen C, Mochly-Rosen D. Opposing effects of delta and xi PKC in ethanol-induced cardioprotection. J Mol Cell Cardiol. 2001;33(3):581–585. - PubMed
-
- Krenz M, Baines CP, Heusch G, Downey JM, Cohen MV. Acute alcohol-induced protection against infarction in rabbit hearts: differences from and similarities to ischemic preconditioning. J Mol Cell Cardiol. 2001;33(11):2015–2022. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

