Polyfluorophore excimers and exciplexes as FRET donors in DNA
- PMID: 19916519
- PMCID: PMC2795122
- DOI: 10.1021/bc9003926
Polyfluorophore excimers and exciplexes as FRET donors in DNA
Abstract
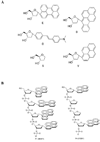
We describe studies aimed at testing whether oligomeric exciplex and excimer fluorophores conjugated to DNA have the potential to act as donors for energy transfer by the Forster mechanism. Oligodeoxyfluorosides (ODFs) are composed of stacked, electronically interacting fluorophores replacing the bases on a DNA scaffold. The monomer chromophores in the twenty tetramer-length ODFs studied here include pyrene (Y), benzopyrene (B), perylene (E), dimethylaminostilbene (D), and a nonfluorescent spacer (S); these are conjugated in varied combinations at the 3' end of a 14mer DNA probe sequence. In the absence of an acceptor chromophore, many of the ODF-DNAs show broad, unstructured long-wavelength emission peaks characteristic of excimer and exciplex excited states, similar to what has been observed for unconjugated ODFs. Although such delocalized excited states have been widely studied, we know of no prior report of their use in FRET. We tested the ability of the twenty ODFs to donate energy to Cy5 and TAMRA dyes conjugated to a complementary strand of DNA, with these acceptors oriented either at the near or far end of the ODF-conjugated probes. Results showed that a number of the ODF fluorophores exhibited relatively efficient energy transfer characteristic of the Forster mechanism, as judged by drops in donor emission quantum yield and fluorescence lifetime, accompanied by increases in intensity of acceptor emission bands. Excimer/exciplex bands in the donors were selectively quenched while shorter-wavelength monomer emission stayed relatively constant, consistent with the notion that the delocalized excited states, rather than individual fluorophores, are the donors. Interestingly, only specific sequences of ODFs were able to act as donors, while others did not, even though their emission wavelengths were similar. The new FRET donors possess large Stokes shifts, which can be beneficial for multiple applications. In addition, all ODFs can be excited at a single wavelength; thus, ODFs may be candidates as "universal FRET donors", thus allowing multicolor FRET of multiple species to be carried out with one excitation.
Figures



Similar articles
-
Polyfluorophore labels on DNA: dramatic sequence dependence of quenching.Chemistry. 2009 Nov 2;15(43):11551-8. doi: 10.1002/chem.200901607. Chemistry. 2009. PMID: 19780115 Free PMC article.
-
Studies of oligodeoxyfluorosides (ODFs) as FRET probes for DNA hybridization.Nucleic Acids Symp Ser (Oxf). 2008;(52):233-4. doi: 10.1093/nass/nrn118. Nucleic Acids Symp Ser (Oxf). 2008. PMID: 18776339
-
An efficient fluorescence resonance energy transfer (FRET) between pyrene and perylene assembled in a DNA duplex and its potential for discriminating single-base changes.Chemistry. 2010 Feb 22;16(8):2479-86. doi: 10.1002/chem.200902078. Chemistry. 2010. PMID: 20066689
-
Fluorescence resonance energy transfer (FRET) and competing processes in donor-acceptor substituted DNA strands: a comparative study of ensemble and single-molecule data.J Biotechnol. 2002 Jan;82(3):211-31. doi: 10.1016/s1389-0352(01)00039-3. J Biotechnol. 2002. PMID: 11999691 Review.
-
Fluorescent DNA base replacements: Reporters and sensors for biological systems.Org Biomol Chem. 2006 Dec 7;4(23):4265-74. doi: 10.1039/b612284c. Epub 2006 Oct 23. Org Biomol Chem. 2006. PMID: 17102869 Review.
Cited by
-
When one plus one does not equal two: fluorescence anisotropy in aggregates and multiply labeled proteins.Biophys J. 2014 Apr 1;106(7):1457-66. doi: 10.1016/j.bpj.2014.02.020. Biophys J. 2014. PMID: 24703307 Free PMC article.
-
DNA-multichromophore systems.Chem Rev. 2012 Jul 11;112(7):4221-45. doi: 10.1021/cr100351g. Epub 2012 Mar 16. Chem Rev. 2012. PMID: 22424059 Free PMC article. Review. No abstract available.
-
Nucleic Acid-Based Nanodevices in Biological Imaging.Annu Rev Biochem. 2016 Jun 2;85:349-73. doi: 10.1146/annurev-biochem-060815-014244. Annu Rev Biochem. 2016. PMID: 27294440 Free PMC article. Review.
-
Protease probes built from DNA: multispectral fluorescent DNA-peptide conjugates as caspase chemosensors.Angew Chem Int Ed Engl. 2011 May 23;50(22):5105-9. doi: 10.1002/anie.201007805. Epub 2011 Mar 31. Angew Chem Int Ed Engl. 2011. PMID: 21455915 Free PMC article. No abstract available.
-
Luminescent Carbon Dot Mimics Assembled on DNA.J Am Chem Soc. 2017 Sep 20;139(37):13147-13155. doi: 10.1021/jacs.7b07420. Epub 2017 Sep 12. J Am Chem Soc. 2017. PMID: 28841010 Free PMC article.
References
-
- Hoeben FJ, Jonkheijm P, Meijer EW, Schenning AP. About supramolecular assemblies of pi-conjugated systems. Chem Rev. 2005;105:1491–1546. - PubMed
-
- Bazan GC. Novel organic materials through control of multichromophore interactions. J Org Chem. 2007;72:8615–8635. - PubMed
-
- Ho HA, Najari A, Leclerc M. Optical detection of DNA and proteins with cationic polythiophenes. Acc Chem Res. 2008;41:168–178. - PubMed
-
- Gensch T, Hofkens J, Heirmann A, Tsuda K, Verheijen W, Vosch T, Christ T, Basche T, Mullen K, De Schryver FC. Fluorescence Detection from Single Dendrimers with Multiple Chromophores. Angew Chem Int Ed Engl. 1999;38:3752–3756. - PubMed
-
- De Schryver FC, Vosch T, Cotlet M, Van der Auweraer M, Mullen K, Hofkens J. Energy dissipation in multichromophoric single dendrimers. Acc Chem Res. 2005;38:514–522. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

