Induced ER chaperones regulate a receptor-like kinase to mediate antiviral innate immune response in plants
- PMID: 19917500
- PMCID: PMC2784700
- DOI: 10.1016/j.chom.2009.10.005
Induced ER chaperones regulate a receptor-like kinase to mediate antiviral innate immune response in plants
Abstract
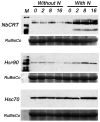
Mounting an effective innate immune response against pathogens requires the rapid and global reprogramming of host cellular processes. Here we employed complementary proteomic methods to identify differentially regulated proteins early during a plant's defense response. Besides defense-related proteins, constituents of the largest category of upregulated proteins were cytoplasmic- and ER-residing molecular chaperones. Investigating the significance of upregulated ER chaperones, we find that silencing of ER-resident protein disulfide isomerases NbERp57 and NbP5 and the calreticulins NbCRT2 and NbCRT3 led to partial loss of N immune receptor-mediated defense against Tobacco mosaic virus (TMV). Furthermore, NbCRT2 and NbCRT3 were required for the expression of a previously uncharacterized induced receptor-like kinase (IRK). IRK is a plasma membrane-localized protein required for N-mediated hypersensitive response, programmed cell death, and resistance to TMV. These data support a model in which ER-resident chaperones are required for the accumulation of membrane-bound or secreted proteins during plant innate immunity.
Figures




References
-
- Austin MJ, Muskett P, Kahn K, Feys BJ, Jones JD, Parker JE. Regulatory role of SGT1 in early R gene-mediated plant defenses. Science. 2002;295:2077–2080. - PubMed
-
- Azevedo C, Sadanandom A, Kitagawa K, Frelaldenhoven A, Shirasu K, Schulze-Lefert P. The Rar1 interactor SGT1, an essential component of R gene-triggered disease resistance. Science. 2002;295:2073–2076. - PubMed
-
- Beachy RN, Murakishi HH. Effect of cycloheximide on tobacco mosaic virus synthesis in callus from hypersensitive tobacco. Virology. 1973;55:320–328. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

