Genetic immunization converts the trypanosoma cruzi B-Cell mitogen proline racemase to an effective immunogen
- PMID: 19917711
- PMCID: PMC2812217
- DOI: 10.1128/IAI.00926-09
Genetic immunization converts the trypanosoma cruzi B-Cell mitogen proline racemase to an effective immunogen
Abstract
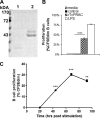
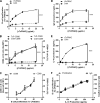
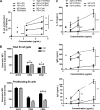
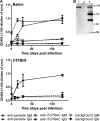
Trypanosoma cruzi is the etiologic agent of Chagas' disease. Acute T. cruzi infection results in polyclonal B-cell activation and delayed specific humoral immunity. T. cruzi proline racemase (TcPRAC), a T. cruzi B-cell mitogen, may contribute to this dysfunctional humoral response. Stimulation of murine splenocytes with recombinant protein (rTcPRAC) induced B-cell proliferation, antibody secretion, interleukin-10 (IL-10) production, and upregulation of CD69 and CD86 on B cells. Marginal zone (MZ) B cells are more responsive to T-cell-independent (TI) rTcPRAC stimulation than are follicular mature (FM) B cells in terms of proliferation, antibody secretion, and IL-10 production. During experimental T. cruzi infection, TcPRAC-specific IgG remained undetectable when responses to other T. cruzi antigens developed. Conversely, intradermal genetic immunization via gene gun (GG) delivered TcPRAC as an immunogen, generating high-titer TcPRAC-specific IgG without B-cell dysfunction. TcPRAC GG immunization led to antigen-specific splenic memory B-cell and bone marrow plasma cell formation. TcPRAC-specific IgG bound mitogenic rTcPRAC, decreasing subsequent B-cell activation. GG immunization with rTcPRAC DNA was nonmitogenic and did not affect the generation of specific IgG to another T. cruzi antigen, complement regulatory protein (CRP). These data demonstrate the utility of genetic immunization for the conversion of a protein mitogen to an effective antigen. Furthermore, coimmunization of TcPRAC with another T. cruzi antigen indicates the usefulness of this approach for multivalent vaccine development.
Figures

Similar articles
-
Genetic immunization elicits antigen-specific protective immune responses and decreases disease severity in Trypanosoma cruzi infection.Infect Immun. 2002 Oct;70(10):5547-55. doi: 10.1128/IAI.70.10.5547-5555.2002. Infect Immun. 2002. PMID: 12228281 Free PMC article.
-
Induction of B- and T-cell responses to cruzipain in the murine model of Trypanosoma cruzi infection.Microbes Infect. 2002 Jul;4(8):805-13. doi: 10.1016/s1286-4579(02)01600-3. Microbes Infect. 2002. PMID: 12270727
-
Specific humoral immunity versus polyclonal B cell activation in Trypanosoma cruzi infection of susceptible and resistant mice.PLoS Negl Trop Dis. 2010 Jul 6;4(7):e733. doi: 10.1371/journal.pntd.0000733. PLoS Negl Trop Dis. 2010. PMID: 20625554 Free PMC article.
-
Parasite polyclonal activators: new targets for vaccination approaches?Int J Parasitol. 2001 May 1;31(5-6):588-91. doi: 10.1016/s0020-7519(01)00171-0. Int J Parasitol. 2001. PMID: 11334946 Review.
-
Trypanosoma cruzi-induced molecular mimicry and Chagas' disease.Curr Top Microbiol Immunol. 2005;296:89-123. doi: 10.1007/3-540-30791-5_6. Curr Top Microbiol Immunol. 2005. PMID: 16323421 Review.
Cited by
-
Developments in the management of Chagas cardiomyopathy.Expert Rev Cardiovasc Ther. 2015 Dec;13(12):1393-409. doi: 10.1586/14779072.2015.1103648. Epub 2015 Oct 23. Expert Rev Cardiovasc Ther. 2015. PMID: 26496376 Free PMC article. Review.
-
Unraveling the Missing Pieces: Exploring the Gaps in Understanding Chagas Cardiomyopathy.Cureus. 2024 Aug 15;16(8):e66955. doi: 10.7759/cureus.66955. eCollection 2024 Aug. Cureus. 2024. PMID: 39280489 Free PMC article. Review.
-
Role of Hormonal Circuitry Upon T Cell Development in Chagas Disease: Possible Implications on T Cell Dysfunctions.Front Endocrinol (Lausanne). 2018 Jun 14;9:334. doi: 10.3389/fendo.2018.00334. eCollection 2018. Front Endocrinol (Lausanne). 2018. PMID: 29963015 Free PMC article. Review.
-
Host-parasite dynamics in Chagas disease from systemic to hyper-local scales.Parasite Immunol. 2021 Feb;43(2):e12786. doi: 10.1111/pim.12786. Epub 2020 Sep 24. Parasite Immunol. 2021. PMID: 32799361 Free PMC article. Review.
-
Combined approaches for drug design points the way to novel proline racemase inhibitor candidates to fight Chagas' disease.PLoS One. 2013 Apr 16;8(4):e60955. doi: 10.1371/journal.pone.0060955. Print 2013. PLoS One. 2013. PMID: 23613764 Free PMC article.
References
-
- Abrahamsohn, I. A., and R. L. Coffman. 1996. Trypanosoma cruzi: IL-10, TNF, IFN-γ, and IL-12 regulate innate and acquired immunity to infection. Exp. Parasitol. 84:231-244. - PubMed
-
- Agematsu, K., H. Nagumo, Y. Oguchi, T. Nakazawa, K. Fukushima, K. Yasui, S. Ito, T. Kobata, C. Morimoto, and A. Komiyama. 1998. Generation of plasma cells from peripheral blood memory B cells: synergistic effect of interleukin-10 and CD27/CD70 interaction. Blood 91:173-180. - PubMed
-
- Beucher, M., W. S. Meira, V. Zegarra, L. M. Galvao, E. Chiari, and K. A. Norris. 2003. Expression and purification of functional, recombinant Trypanosoma cruzi complement regulatory protein. Protein Expr. Purif. 27:19-26. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

