MPEP reduces seizure severity in Fmr-1 KO mice over expressing human Abeta
- PMID: 19918329
- PMCID: PMC2776265
MPEP reduces seizure severity in Fmr-1 KO mice over expressing human Abeta
Abstract
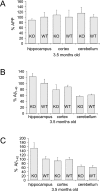
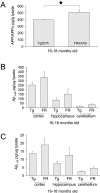
Metabotropic glutamate receptor 5 (mGluR(5)) regulates the translation of amyloid precursor protein (APP) mRNA. Under resting conditions, mRNA is bound to and translationally repressed by the fragile X mental retardation protein (FMRP). Upon group 1 mGluR activation, FMRP dissociates from the mRNA and translation ensues. APP levels are elevated in the dendrites of primary neuronal cultures as well as in synaptoneurosomes (SN) prepared from embryonic and juvenile fmr-1 knockout (KO) mice, respectively. In order to study the effects of APP and its proteolytic product Abeta on Fragile X syndrome (FXS) phenotypes, we created a novel mouse model (FRAXAD) that over-expresses human APPSwe/Abeta in an fmr-1 KO background. Herein, we assess (1) human APP(Swe) and Abeta levels as a function of age in FRAXAD mice, and (2) seizure susceptibility to pentylenetetrazol (PTZ) after mGluR(5) blockade. PTZ-induced seizure severity is decreased in FRAXAD mice pre-treated with the mGluR(5) antagonist MPEP. These data suggest that Abeta contributes to seizure incidence and may be an appropriate therapeutic target to lessen seizure pathology in FXS, Alzheimer's disease (AD) and Down syndrome (DS) patients.
Keywords: APP; FMRP; FRAXAD; PTZ; Tg2576; seizure; β-amyloid.
Figures


Similar articles
-
Fragile X and APP: a Decade in Review, a Vision for the Future.Mol Neurobiol. 2019 Jun;56(6):3904-3921. doi: 10.1007/s12035-018-1344-x. Epub 2018 Sep 17. Mol Neurobiol. 2019. PMID: 30225775 Free PMC article. Review.
-
Seizure susceptibility and mortality in mice that over-express amyloid precursor protein.Int J Clin Exp Pathol. 2008 Jan 1;1(2):157-68. Int J Clin Exp Pathol. 2008. PMID: 18784809 Free PMC article.
-
Reversal of fragile X phenotypes by manipulation of AβPP/Aβ levels in Fmr1KO mice.PLoS One. 2011;6(10):e26549. doi: 10.1371/journal.pone.0026549. Epub 2011 Oct 26. PLoS One. 2011. PMID: 22046307 Free PMC article.
-
FMRP mediates mGluR5-dependent translation of amyloid precursor protein.PLoS Biol. 2007 Mar;5(3):e52. doi: 10.1371/journal.pbio.0050052. PLoS Biol. 2007. PMID: 17298186 Free PMC article.
-
Autism, Alzheimer disease, and fragile X: APP, FMRP, and mGluR5 are molecular links.Neurology. 2011 Apr 12;76(15):1344-52. doi: 10.1212/WNL.0b013e3182166dc7. Neurology. 2011. PMID: 21482951 Free PMC article. Review.
Cited by
-
Targeted pharmacological treatment of autism spectrum disorders: fragile X and Rett syndromes.Front Cell Neurosci. 2015 Feb 26;9:55. doi: 10.3389/fncel.2015.00055. eCollection 2015. Front Cell Neurosci. 2015. PMID: 25767435 Free PMC article. Review.
-
Early-Onset Network Hyperexcitability in Presymptomatic Alzheimer's Disease Transgenic Mice Is Suppressed by Passive Immunization with Anti-Human APP/Aβ Antibody and by mGluR5 Blockade.Front Aging Neurosci. 2017 Mar 24;9:71. doi: 10.3389/fnagi.2017.00071. eCollection 2017. Front Aging Neurosci. 2017. PMID: 28392767 Free PMC article.
-
Taking STEPs forward to understand fragile X syndrome.Results Probl Cell Differ. 2012;54:223-41. doi: 10.1007/978-3-642-21649-7_12. Results Probl Cell Differ. 2012. PMID: 22009355 Free PMC article. Review.
-
mRNA and protein expression for novel GABAA receptors θ and ρ2 are altered in schizophrenia and mood disorders; relevance to FMRP-mGluR5 signaling pathway.Transl Psychiatry. 2013 Jun 18;3(6):e271. doi: 10.1038/tp.2013.46. Transl Psychiatry. 2013. PMID: 23778581 Free PMC article.
-
Fragile X and APP: a Decade in Review, a Vision for the Future.Mol Neurobiol. 2019 Jun;56(6):3904-3921. doi: 10.1007/s12035-018-1344-x. Epub 2018 Sep 17. Mol Neurobiol. 2019. PMID: 30225775 Free PMC article. Review.
References
-
- Hagerman RJ, Hagerman PJ. Physical and Behavioral Phenotype. Baltimore: John Hopkins University Press; 2002.
-
- Velez L, Selwa LM. Seizure disorders in the elderly. Am Fam Physician. 2003;67:325–332. - PubMed
-
- Risse SC, Lampe TH, Bird TD, Nochlin D, Sumi SM, Keenan T, Cubberley L, Peskind E, Raskind MA. Myoclonus, seizures, and paratonia in alzheimer disease. Alzheimer Dis Assoc Disord. 1990;4:217–225. - PubMed
-
- Menendez M. Down syndrome, alzheimer's disease and seizures. Brain Dev. 2005;27:246–252. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
