Aggrecanolysis and in vitro matrix degradation in the immature bovine meniscus: mechanisms and functional implications
- PMID: 19919704
- PMCID: PMC3003508
- DOI: 10.1186/ar2862
Aggrecanolysis and in vitro matrix degradation in the immature bovine meniscus: mechanisms and functional implications
Abstract
Introduction: Little is known about endogenous or cytokine-stimulated aggrecan catabolism in the meniscal fibrocartilage of the knee. The objectives of this study were to characterize the structure, distribution, and processing of aggrecan in menisci from immature bovines, and to identify mechanisms of extracellular matrix degradation that lead to changes in the mechanical properties of meniscal fibrocartilage.
Methods: Aggrecanase activity in the native immature bovine meniscus was examined by immunolocalization of the aggrecan NITEGE neoepitope. To investigate mechanisms of cytokine-induced aggrecan catabolism in this tissue, explants were treated with interleukin-1alpha (IL-1) in the absence or presence of selective or broad spectrum metalloproteinase inhibitors. The sulfated glycosaminoglycan (sGAG) and collagen contents of explants and culture media were quantified by biochemical methods, and aggrecan catabolism was examined by Western analysis of aggrecan fragments. The mechanical properties of explants were determined by dynamic compression and shear tests.
Results: The aggrecanase-generated NITEGE neoepitope was preferentially localized in the middle and outer regions of freshly isolated immature bovine menisci, where sGAG density was lowest and blood vessels were present. In vitro treatment of explants with IL-1 triggered the accumulation of NITEGE in the inner and middle regions. Middle region explants stimulated with IL-1 exhibited substantial decreases in sGAG content, collagen content, and mechanical properties. A broad spectrum metalloproteinase inhibitor significantly reduced sGAG loss, abrogated collagen degradation, and preserved tissue mechanical properties. In contrast, an inhibitor selective for ADAMTS-4 and ADAMTS-5 was least effective at blocking IL-1-induced matrix catabolism and loss of mechanical properties.
Conclusions: Aggrecanase-mediated aggrecanolysis, typical of degenerative articular cartilage, may play a physiologic role in the development of the immature bovine meniscus. IL-1-induced release of sGAG and loss of mechanical properties can be ascribed primarily to the activity of MMPs or aggrecanases other than ADAMTS-4 and ADAMTS-5. These results may have implications for the clinical management of osteoarthritis.
Figures



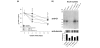
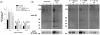
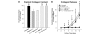
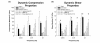
Similar articles
-
Interleukin-1alpha treatment of meniscal explants stimulates the production and release of aggrecanase-generated, GAG-substituted aggrecan products and also the release of pre-formed, aggrecanase-generated G1 and m-calpain-generated G1-G2.Cell Tissue Res. 2010 Apr;340(1):179-88. doi: 10.1007/s00441-010-0941-4. Epub 2010 Mar 9. Cell Tissue Res. 2010. PMID: 20217136
-
Release of hyaluronan and hyaladherins (aggrecan G1 domain and link proteins) from articular cartilage exposed to ADAMTS-4 (aggrecanase 1) or ADAMTS-5 (aggrecanase 2).Arthritis Rheum. 2004 Sep;50(9):2839-48. doi: 10.1002/art.20496. Arthritis Rheum. 2004. PMID: 15457452
-
Nitric oxide enhances aggrecan degradation by aggrecanase in response to TNF-alpha but not IL-1beta treatment at a post-transcriptional level in bovine cartilage explants.Osteoarthritis Cartilage. 2008 Apr;16(4):489-97. doi: 10.1016/j.joca.2007.07.015. Epub 2007 Oct 10. Osteoarthritis Cartilage. 2008. PMID: 17923423 Free PMC article.
-
Mechanisms involved in cartilage proteoglycan catabolism.Matrix Biol. 2000 Aug;19(4):333-44. doi: 10.1016/s0945-053x(00)00078-0. Matrix Biol. 2000. PMID: 10963994 Review.
-
Aggrecanase and aggrecan degradation in osteoarthritis: a review.J Int Med Res. 2008 Nov-Dec;36(6):1149-60. doi: 10.1177/147323000803600601. J Int Med Res. 2008. PMID: 19094423 Review.
Cited by
-
Explant models for meniscus metabolism, injury, repair, and healing.Connect Tissue Res. 2020 May-Jul;61(3-4):292-303. doi: 10.1080/03008207.2019.1702031. Epub 2019 Dec 16. Connect Tissue Res. 2020. PMID: 31842590 Free PMC article. Review.
-
Interleukin-1, tumor necrosis factor-alpha, and transforming growth factor-beta 1 and integrative meniscal repair: influences on meniscal cell proliferation and migration.Arthritis Res Ther. 2011;13(6):R187. doi: 10.1186/ar3515. Epub 2011 Nov 16. Arthritis Res Ther. 2011. PMID: 22087734 Free PMC article.
-
Meniscus is more susceptible than cartilage to catabolic and anti-anabolic effects of adipokines.Osteoarthritis Cartilage. 2015 Sep;23(9):1551-62. doi: 10.1016/j.joca.2015.04.014. Epub 2015 Apr 23. Osteoarthritis Cartilage. 2015. PMID: 25917638 Free PMC article.
-
Dynamic loading enhances integrative meniscal repair in the presence of interleukin-1.Osteoarthritis Cartilage. 2010 Jun;18(6):830-8. doi: 10.1016/j.joca.2010.02.009. Epub 2010 Feb 14. Osteoarthritis Cartilage. 2010. PMID: 20202487 Free PMC article.
-
Electrical stimulation enhances cell migration and integrative repair in the meniscus.Sci Rep. 2014 Jan 14;4:3674. doi: 10.1038/srep03674. Sci Rep. 2014. PMID: 24419206 Free PMC article.
References
-
- McDevitt CA, Webber RJ. The ultrastructure and biochemistry of meniscal cartilage. Clin Orthop Relat Res. 1990;252:8–18. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

