Reciprocal negative regulation of PDK1 and ASK1 signaling by direct interaction and phosphorylation
- PMID: 19920149
- PMCID: PMC2807298
- DOI: 10.1074/jbc.M109.064295
Reciprocal negative regulation of PDK1 and ASK1 signaling by direct interaction and phosphorylation
Abstract
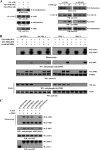
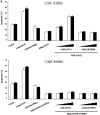
Cell survival and death-inducing signals are tightly associated with each other, and the decision as to whether a cell survives or dies is determined by controlling the relationship between these signals. However, the mechanism underlying the reciprocal regulation of such signals remains unclear. In this study, we reveal a functional association between PDK1 (3-phosphoinositide-dependent protein kinase 1), a critical mediator of cell survival, and ASK1 (apoptosis signal-regulating kinase 1), an apoptotic stress-activated MAPKKK. The physical association between PDK1 and ASK1 is mediated through the pleckstrin homology domain of PDK1 and the C-terminal regulatory domain of ASK1 and is decreased by ASK1-activating stimuli, such as H(2)O(2), tumor necrosis factor alpha, thapsigargin, and ionomycin, as well as insulin, a PDK1 stimulator. Wild-type PDK1, but not kinase-dead PDK1, negatively regulates ASK1 activity by phosphorylating Ser(967), a binding site for 14-3-3 protein, on ASK1. PDK1 functionally suppresses ASK1-mediated AP-1 transactivation and H(2)O(2)-mediated apoptosis in a kinase-dependent manner. On the other hand, ASK1 has been shown to inhibit PDK1 functions, including PDK1-mediated regulation of apoptosis and cell growth, by phosphorylating PDK1 at Ser(394) and Ser(398), indicating that these putative phosphorylation sites are involved in the negative regulation of PDK1 activity. These results provide evidence that PDK1 and ASK1 directly interact and phosphorylate each other and act as negative regulators of their respective kinases in resting cells.
Figures












References
-
- Brunet A., Bonni A., Zigmond M. J., Lin M. Z., Juo P., Hu L. S., Anderson M. J., Arden K. C., Blenis J., Greenberg M. E. (1999) Cell 96, 857–868 - PubMed
-
- Cardone M. H., Roy N., Stennicke H. R., Salvesen G. S., Franke T. F., Stanbridge E., Frisch S., Reed J. C. (1998) Science 282, 1318–1321 - PubMed
-
- Cross D. A., Alessi D. R., Cohen P., Andjelkovich M., Hemmings B. A. (1995) Nature 378, 785–789 - PubMed
-
- Datta S. R., Dudek H., Tao X., Masters S., Fu H., Gotoh Y., Greenberg M. E. (1997) Cell 91, 231–241 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

