Antidiabetic Bis-Maltolato-OxoVanadium(IV): conversion of inactive trans- to bioactive cis-BMOV for possible binding to target PTP-1B
- PMID: 19920909
- PMCID: PMC2761195
Antidiabetic Bis-Maltolato-OxoVanadium(IV): conversion of inactive trans- to bioactive cis-BMOV for possible binding to target PTP-1B
Abstract
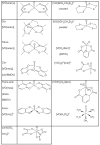
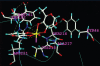
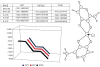
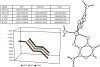
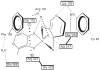
The postulated transition of Bis-Maltolato-OxoVanadium(IV) (BMOV) from its inactive trans- into its cis-aquo-BMOV isomeric form in solution was simulated by means of computational molecular modeling. The rotational barrier was calculated with DFT - B3LYP under a stepwise optimization protocol with STO-3G, 3-21G, 3-21G*, and 6-31G ab initio basis sets. Our computed results are consistent with reports on the putative molecular mechanism of BMOV triggering the insulin-like cellular response (insulin mimetic) as a potent inhibitor of the protein tyrosine phosphatase-1B (PTP-1B). Initially, trans-BMOV is present in its solid dosage form but in aqueous solution, and during oral administration, it is readily converted into a mixture of "open-type" and "closed-type" complexes of cis-aquo-BMOV under equilibrium conditions. However, in the same measure as the "closed-type" complex binds to the cytosolic PTP-1B, it disappears from solution, and the equilibrium shifts towards the "closed-type" species. In full accordance, the computed binding mode of cis-BMOV is energetically favored over sterically hindered trans-BMOV. In view of our earlier report on prodrug hypothesis of vanadium organic compounds the present results suggest that cis-BMOV is the bioactive species.
Keywords: PTP-1B; computational quantum chemistry; diabetes mellitus; molecular modeling; vanadium compounds.
Figures
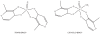

Similar articles
-
Inhibition of protein tyrosine phosphatase 1B and alkaline phosphatase by bis(maltolato)oxovanadium (IV).J Inorg Biochem. 2008 Oct;102(10):1846-53. doi: 10.1016/j.jinorgbio.2008.06.007. Epub 2008 Jun 20. J Inorg Biochem. 2008. PMID: 18728000
-
Comparison of the glucose-lowering properties of vanadyl sulfate and bis(maltolato)oxovanadium(IV) following acute and chronic administration.Can J Physiol Pharmacol. 1995 Jan;73(1):55-64. doi: 10.1139/y95-008. Can J Physiol Pharmacol. 1995. PMID: 7600453
-
New insights into the interactions of serum proteins with bis(maltolato)oxovanadium(IV): transport and biotransformation of insulin-enhancing vanadium pharmaceuticals.J Am Chem Soc. 2005 Apr 13;127(14):5104-15. doi: 10.1021/ja043944n. J Am Chem Soc. 2005. PMID: 15810845
-
Bis(maltolato)-oxovanadium (IV)-induced phosphorylation of PKB, GSK-3 and FOXO1 contributes to its glucoregulatory responses (review).Int J Mol Med. 2009 Sep;24(3):303-9. doi: 10.3892/ijmm_00000233. Int J Mol Med. 2009. PMID: 19639221 Review.
-
Increased potency of vanadium using organic ligands.Mol Cell Biochem. 1995 Dec 6-20;153(1-2):175-80. doi: 10.1007/BF01075935. Mol Cell Biochem. 1995. PMID: 8927036 Review.
Cited by
-
Are vanadium complexes druggable against the main protease Mpro of SARS-CoV-2? - A computational approach.Inorganica Chim Acta. 2021 May 1;519:120287. doi: 10.1016/j.ica.2021.120287. Epub 2021 Feb 11. Inorganica Chim Acta. 2021. PMID: 33589845 Free PMC article.
-
Why Antidiabetic Vanadium Complexes are Not in the Pipeline of "Big Pharma" Drug Research? A Critical Review.Curr Med Chem. 2016;23(25):2874-2891. doi: 10.2174/0929867323666160321121138. Curr Med Chem. 2016. PMID: 26997154 Free PMC article. Review.
-
Sodium Orthovanadate Changes Fatty Acid Composition and Increased Expression of Stearoyl-Coenzyme A Desaturase in THP-1 Macrophages.Biol Trace Elem Res. 2020 Jan;193(1):152-161. doi: 10.1007/s12011-019-01699-2. Epub 2019 Mar 29. Biol Trace Elem Res. 2020. PMID: 30927246 Free PMC article.
-
Synthesis, Characterization, and In Vitro Insulin-Mimetic Activity Evaluation of Valine Schiff Base Coordination Compounds of Oxidovanadium(V).Biomedicines. 2021 May 17;9(5):562. doi: 10.3390/biomedicines9050562. Biomedicines. 2021. PMID: 34067862 Free PMC article.
-
Exploring the Biological Effects of Anti-Diabetic Vanadium Compounds in the Liver, Heart and Brain.Diabetes Metab Syndr Obes. 2024 Sep 4;17:3267-3278. doi: 10.2147/DMSO.S417700. eCollection 2024. Diabetes Metab Syndr Obes. 2024. PMID: 39247428 Free PMC article. Review.
References
-
- Adachi Y, Yoshikawa Y, Yoshida J, et al. Improvement of diabetes, obesity and hypertension in type 2 diabetic KKA(y) mice by bis(alli-xinato)oxovanadium(IV) complex. Biochem Biophys Res Commun. 2006;345:945–50. - PubMed
-
- ArgusLab. ArgusLab 4.0.1. Mark A. Thompson. Planaria Software LLC; Seattle, WA: 2005.
-
- Atkins PW, Friedman RS. Molecular Quantum Mechanics. Oxford University Press; Oxford, New York, Toronto: 2003.
-
- Aznar CP, Deligiannakis Y, Tolis EJ, et al. ESE-ENDOR study and DFT calculations on oxovanadium compounds: Effect of axial anionic ligands on the 51V nuclear quadrupolar coupling constant. J Phys Chem. 2004;108:4310–21.
-
- Barford D, Flint AJ, Tonks NK. Crystal structure of human protein tyrosine phosphatase 1B. Science. 1994;263(5152):1397–404. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

