Orexin/hypocretin and histamine: distinct roles in the control of wakefulness demonstrated using knock-out mouse models
- PMID: 19923277
- PMCID: PMC2802289
- DOI: 10.1523/JNEUROSCI.2604-09.2009
Orexin/hypocretin and histamine: distinct roles in the control of wakefulness demonstrated using knock-out mouse models
Abstract
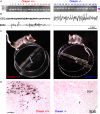
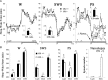
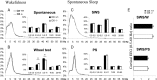
To determine the respective role played by orexin/hypocretin and histamine (HA) neurons in maintaining wakefulness (W), we characterized the behavioral and sleep-wake phenotypes of orexin (Ox) knock-out (-/-) mice and compared them with those of histidine-decarboxylase (HDC, HA-synthesizing enzyme)-/- mice. While both mouse strains displayed sleep fragmentation and increased paradoxical sleep (PS), they presented a number of marked differences: (1) the PS increase in HDC(-/-) mice was seen during lightness, whereas that in Ox(-/-) mice occurred during darkness; (2) contrary to HDC(-/-), Ox(-/-) mice had no W deficiency around lights-off, nor an abnormal EEG and responded to a new environment with increased W; (3) only Ox(-/-), but not HDC(-/-) mice, displayed narcolepsy and deficient W when faced with motor challenge. Thus, when placed on a wheel, wild-type (WT), but not littermate Ox(-/-) mice, voluntarily spent their time in turning it and as a result, remained highly awake; this was accompanied by dense c-fos expression in many areas of their brains, including Ox neurons in the dorsolateral hypothalamus. The W and motor deficiency of Ox(-/-) mice was due to the absence of Ox because intraventricular dosing of orexin-A restored their W amount and motor performance whereas SB-334867 (Ox1-receptor antagonist, i.p.) impaired W and locomotion of WT mice during the test. These data indicate that Ox, but not HA, promotes W through enhanced locomotion and suggest that HA and Ox neurons exert a distinct, but complementary and synergistic control of W: the neuropeptide being more involved in its behavioral aspects, whereas the amine is mainly responsible for its qualitative cognitive aspects and cortical EEG activation.
Figures





Comment in
-
Histamine and orexin in the control of arousal, locomotion, and motivation.J Neurosci. 2010 Feb 24;30(8):2810-1. doi: 10.1523/JNEUROSCI.0045-10.2010. J Neurosci. 2010. PMID: 20181578 Free PMC article. Review. No abstract available.
References
-
- Anaclet C, Parmentier R, Guidon G, Buda C, Sastre JP, Haas HL, Lin JS. Respective role of histamine and orexin neurons in sleep-wake control. Sleep. 2007;30(Suppl):A24.
-
- Bayer L, Eggermann E, Serafin M, Saint-Mleux B, Machard D, Jones B, Mühlethaler M. Orexins (hypocretins) directly excite tuberomammillary neurons. Eur J Neurosci. 2001;14:1571–1575. - PubMed
-
- Chemelli RM, Willie JT, Sinton CM, Elmquist JK, Scammell T, Lee C, Richardson JA, Williams SC, Xiong Y, Kisanuki Y, Fitch TE, Nakazato M, Hammer RE, Saper CB, Yanagisawa M. Narcolepsy in orexin knockout mice: molecular genetics of sleep regulation. Cell. 1999;98:437–451. - PubMed
-
- de Lecea L, Kilduff TS, Peyron C, Gao X, Foye PE, Danielson PE, Fukuhara C, Battenberg EL, Gautvik VT, Bartlett FS, 2nd, Frankel WN, van den Pol AN, Bloom FE, Gautvik KM, Sutcliffe JG. The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc Natl Acad Sci U S A. 1998;95:322–327. - PMC - PubMed
-
- Duxon MS, Stretton J, Starr K, Jones DN, Holland V, Riley G, Jerman J, Brough S, Smart D, Johns A, Chan W, Porter RA, Upton N. Evidence that orexin-A-evoked grooming in the rat is mediated by orexin-1 (OX1) receptors, with downstream 5-HT2C receptor involvement. Psychopharmacology (Berl) 2001;153:203–209. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
