Divergent pathways mediate spine alterations and cell death induced by amyloid-beta, wild-type tau, and R406W tau
- PMID: 19923278
- PMCID: PMC6665808
- DOI: 10.1523/JNEUROSCI.3590-09.2009
Divergent pathways mediate spine alterations and cell death induced by amyloid-beta, wild-type tau, and R406W tau
Abstract
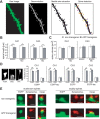
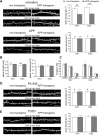
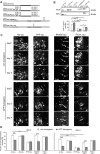
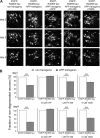
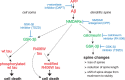
Alzheimer's disease is characterized by synaptic alterations and neurodegeneration. Histopathological hallmarks represent amyloid plaques composed of amyloid-beta (Abeta) and neurofibrillary tangles containing hyperphosphorylated tau. To determine whether synaptic changes and neurodegeneration share common pathways, we established an ex vivo model using organotypic hippocampal slice cultures from amyloid precursor protein transgenic mice combined with virus-mediated expression of EGFP-tagged tau constructs. Confocal high-resolution imaging, algorithm-based evaluation of spines, and live imaging were used to determine spine changes and neurodegeneration. We report that Abeta but not tau induces spine loss and shifts spine shape from mushroom to stubby through a mechanism involving NMDA receptor (NMDAR), calcineurin, and GSK-3beta activation. In contrast, Abeta alone does not cause neurodegeneration but induces toxicity through phosphorylation of wild-type (wt) tau in an NMDAR-dependent pathway. We show that GSK-3beta levels are elevated in APP transgenic cultures and that inhibiting GSK-3beta activity or use of phosphorylation-blocking tau mutations prevented Abeta-induced toxicity of tau. FTDP-17 tau mutants are differentially affected by Abeta. While R406W tau shows increased toxicity in the presence of Abeta, no change is observed with P301L tau. While blocking NMDAR activity abolishes toxicity of both wt and R406W tau, the inhibition of GSK-3beta only protects against toxicity of wt tau but not of R406W tau induced by Abeta. Tau aggregation does not correlate with toxicity. We propose that Abeta-induced spine pathology and tau-dependent neurodegeneration are mediated by divergent pathways downstream of NMDAR activation and suggest that Abeta affects wt and R406W tau toxicity by different pathways downstream of NMDAR activity.
Figures



References
-
- Blanchard V, Moussaoui S, Czech C, Touchet N, Bonici B, Planche M, Canton T, Jedidi I, Gohin M, Wirths O, Bayer TA, Langui D, Duyckaerts C, Tremp G, Pradier L. Time sequence of maturation of dystrophic neurites associated with Abeta deposits in APP/PS1 transgenic mice. Exp Neurol. 2003;184:247–263. - PubMed
-
- Blurton-Jones M, Laferla FM. Pathways by which Abeta facilitates tau pathology. Curr Alzheimer Res. 2006;3:437–448. - PubMed
-
- Bourne J, Harris KM. Do thin spines learn to be mushroom spines that remember? Curr Opin Neurobiol. 2007;17:381–386. - PubMed
-
- Braak H, Braak E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991;82:239–259. - PubMed
-
- Chen P, Gu Z, Liu W, Yan Z. Glycogen synthase kinase 3 regulates N-methyl-D-aspartate receptor channel trafficking and function in cortical neurons. Mol Pharmacol. 2007;72:40–51. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
