The distal pocket histidine residue in horse heart myoglobin directs the O-binding mode of nitrite to the heme iron
- PMID: 19924902
- PMCID: PMC2824769
- DOI: 10.1021/ja904726q
The distal pocket histidine residue in horse heart myoglobin directs the O-binding mode of nitrite to the heme iron
Abstract
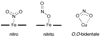
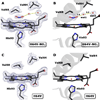
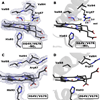
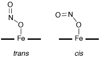
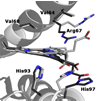
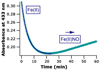
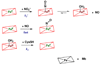
It is now well-established that mammalian heme proteins are reactive with various nitrogen oxide species and that these reactions may play significant roles in mammalian physiology. For example, the ferrous heme protein myoglobin (Mb) has been shown to reduce nitrite (NO(2)(-)) to nitric oxide (NO) under hypoxic conditions. We demonstrate here that the distal pocket histidine residue (His64) of horse heart metMb(III) (i.e., ferric Mb(III)) has marked effects on the mode of nitrite ion coordination to the iron center. X-ray crystal structures were determined for the mutant proteins metMb(III) H64V (2.0 A resolution) and its nitrite ion adduct metMb(III) H64V-nitrite (1.95 A resolution), and metMb(III) H64V/V67R (1.9 A resolution) and its nitrite ion adduct metMb(III) H64V/V67R-nitrite (2.0 A resolution). These are compared to the known structures of wild-type (wt) hh metMb(III) and its nitrite ion adduct hh metMb(III)-nitrite, which binds NO(2)(-) via an O-atom in a trans-FeONO configuration. Unlike wt metMb(III), no axial H(2)O is evident in either of the metMb(III) mutant structures. In the ferric H64V-nitrite structure, replacement of the distal His residue with Val alters the binding mode of nitrite from the nitrito (O-binding) form in the wild-type protein to a weakly bound nitro (N-binding) form. Reintroducing a H-bonding residue in the H64V/V67R double mutant restores the O-binding mode of nitrite. We have also examined the effects of these mutations on reactivities of the metMb(III)s with cysteine as a reducing agent and of the (ferrous) Mb(II)s with nitrite ion under anaerobic conditions. The Mb(II)s were generated by reduction of the Mb(III) precursors in a second-order reaction with cysteine, the rate constants for this step following the order H64V/V67R > H64V >> wt. The rate constants for the oxidation of the Mb(II)s by nitrite (giving NO as the other product) follow the order wt > H64V/V67R >> H64V and suggest a significant role of the distal pocket H-bonding residue in nitrite reduction.
Figures

References
-
- Braida W, Ong SK. Water, Air, and Soil Pollution. 2000;118:13–26.
-
- Averill BA. Chem. Rev. 1996;96:2951–2964. - PubMed
-
- Eady RR, Hasnain SS. Denitrification. In: Que L Jr, Tolman WB, editors. Comprehensive Coordination Chemistry II. Vol. 8. San Diego, CA: Elsevier; 2004. pp. 759–786.
-
- Wasser IM, de Vries S, Moenne-Loccoz P, Schroder I, Karlin KD. Chem. Rev. 2002;102:1201–1234. - PubMed
-
- Tavares P, Pereira AS, Moura JJG. J. Inorg. Biochem. 2006;100:2087–2100. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

