Identification of a single peridinin sensing Chl-a excitation in reconstituted PCP by crystallography and spectroscopy
- PMID: 19934052
- PMCID: PMC2791617
- DOI: 10.1073/pnas.0908938106
Identification of a single peridinin sensing Chl-a excitation in reconstituted PCP by crystallography and spectroscopy
Abstract
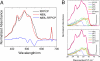
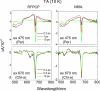
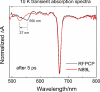
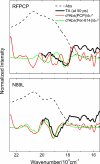
The peridinin-chlorophyll a-protein (PCP) of dinoflagellates is unique among the large variety of natural photosynthetic light-harvesting systems. In contrast to other chlorophyll protein complexes, the soluble PCP is located in the thylakoid lumen, and the carotenoid pigments outnumber the chlorophylls. The structure of the PCP complex consists of two symmetric domains, each with a central chlorophyll a (Chl-a) surrounded by four peridinin molecules. The protein provides distinctive surroundings for the pigment molecules, and in PCP, the specific environment around each peridinin results in overlapping spectral line shapes, suggestive of different functions within the protein. One particular Per, Per-614, is hypothesized to show the strongest electronic interaction with the central Chl-a. We have performed an in vitro reconstitution of pigments into recombinant PCP apo-protein (RFPCP) and into a mutated protein with an altered environment near Per-614. Steady-state and transient optical spectroscopic experiments comparing the RFPCP complex with the reconstituted mutant protein identify specific amino acid-induced spectral shifts. The spectroscopic assignments are reinforced by a determination of the structures of both RFPCP and the mutant by x-ray crystallography to a resolution better than 1.5 A. RFPCP and mutated RFPCP are unique in representing crystal structures of in vitro reconstituted light-harvesting pigment-protein complexes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
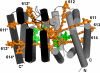
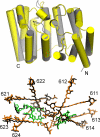
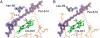
Similar articles
-
X-ray structures of the peridinin-chlorophyll-protein reconstituted with different chlorophylls.FEBS Lett. 2010 Mar 5;584(5):973-8. doi: 10.1016/j.febslet.2010.01.041. Epub 2010 Jan 25. FEBS Lett. 2010. PMID: 20102711
-
Reconstitution of the peridinin-chlorophyll a protein (PCP): evidence for functional flexibility in chlorophyll binding.Photosynth Res. 2005 Nov;86(1-2):229-40. doi: 10.1007/s11120-005-2067-1. Photosynth Res. 2005. PMID: 16172941
-
Structural and spectroscopic characterization of the peridinin-chlorophyll a-protein (PCP) complex from Heterocapsa pygmaea (HPPCP).Biochim Biophys Acta Bioenerg. 2025 Jan 1;1866(1):149510. doi: 10.1016/j.bbabio.2024.149510. Epub 2024 Sep 24. Biochim Biophys Acta Bioenerg. 2025. PMID: 39321862
-
Spectroscopy of the peridinin-chlorophyll-a protein: insight into light-harvesting strategy of marine algae.Arch Biochem Biophys. 2007 Feb 15;458(2):111-20. doi: 10.1016/j.abb.2006.10.006. Epub 2006 Oct 27. Arch Biochem Biophys. 2007. PMID: 17098207 Review.
-
Structure and function of native and refolded peridinin-chlorophyll-proteins from dinoflagellates.Eur J Cell Biol. 2010 Dec;89(12):990-7. doi: 10.1016/j.ejcb.2010.08.004. Epub 2010 Sep 16. Eur J Cell Biol. 2010. PMID: 20846743 Review.
Cited by
-
The nature of the intramolecular charge transfer state in peridinin.Biophys J. 2013 Mar 19;104(6):1314-25. doi: 10.1016/j.bpj.2013.01.045. Epub 2013 Mar 19. Biophys J. 2013. PMID: 23528091 Free PMC article.
-
Carotenoid-Chlorophyll Interactions in a Photosynthetic Antenna Protein: A Supramolecular QM/MM Approach.Molecules. 2018 Oct 10;23(10):2589. doi: 10.3390/molecules23102589. Molecules. 2018. PMID: 30308965 Free PMC article.
-
The Energy Transfer Yield between Carotenoids and Chlorophylls in Peridinin Chlorophyll a Protein Is Robust against Mutations.Int J Mol Sci. 2022 May 3;23(9):5067. doi: 10.3390/ijms23095067. Int J Mol Sci. 2022. PMID: 35563456 Free PMC article.
-
The unique photophysical properties of the Peridinin-Chlorophyll-α-Protein.Curr Protein Pept Sci. 2014;15(4):332-50. doi: 10.2174/1389203715666140327111139. Curr Protein Pept Sci. 2014. PMID: 24678668 Free PMC article. Review.
-
Photoinduced chlorophyll charge transfer state identified in the light-harvesting complex II from a marine green alga Bryopsis corticulans.iScience. 2022 Dec 7;26(1):105761. doi: 10.1016/j.isci.2022.105761. eCollection 2023 Jan 20. iScience. 2022. PMID: 36594012 Free PMC article.
References
-
- Frank HA, Young AJ, Britton G, Cogdell RJ, editors. The Photochemistry of Carotenoids. Dordrecht: Kluwer Academic Publishers; 1999.
-
- Green BR, Parson WW. Advances in Photosynthesis and Respiration Vol. 13: Light Harvesting Antennas. 1st Ed. Dordrecht: Kluwer academic publishers; 2003.
-
- Cogdell RJ, Gall A, Köhler J. The architecture and function of the light-harvesting apparatus of purple bacteria: From single molecules to in vivo membranes. Q Rev Biophys. 2006;39:227–324. - PubMed
-
- Desamero RZB, et al. Mechanism of energy transfer from carotenoids to bacteriochlorophyll: Light-harvesting by carotenoids having different extents of π-electron conjugation incorporated into the B850 antenna complex from the carotenoidless bacterium rhodobacter sphaeroides R-26.1. J Phys Chem B. 1998;102:8151–8162.
-
- Paulsen H, Hobe S. Pigment-binding properties of mutant light-harvesting chlorophyll-a/b-binding protein. Eur J Biochem. 1992;205:71–76. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

