Tobacco smoke exposure induces nicotine dependence in rats
- PMID: 19936715
- PMCID: PMC3586198
- DOI: 10.1007/s00213-009-1716-z
Tobacco smoke exposure induces nicotine dependence in rats
Abstract
Rationale: Tobacco smoke contains nicotine and many other compounds that act in concert on the brain reward system. Therefore, animal models are needed that allow the investigation of chronic exposure to the full spectrum of tobacco smoke constituents.
Objectives: The aim of these studies was to investigate if exposure to tobacco smoke leads to nicotine dependence in rats.
Methods: The intracranial self-stimulation procedure was used to assess the negative affective aspects of nicotine withdrawal. Somatic signs were recorded from a checklist of nicotine abstinence signs. Nicotine self-administration sessions were conducted to investigate if tobacco smoke exposure affects the motivation to self-administer nicotine. Nicotinic receptor autoradiography was used to investigate if exposure to tobacco smoke affects central alpha7 nicotinic acetylcholine receptor (nAChR) and non-alpha7 nAChR levels (primarily alpha4beta2 nAChRs).
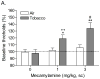
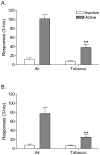
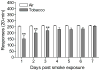
Results: The nAChR antagonist mecamylamine dose-dependently elevated the brain reward thresholds of the rats exposed to tobacco smoke and did not affect the brain reward thresholds of the untreated control rats. Furthermore, mecamylamine induced more somatic withdrawal signs in the smoke-exposed rats than in the control rats. Nicotine self-administration was decreased 1 day after the last tobacco smoke exposure sessions and was returned to control levels 5 days later. Tobacco smoke exposure increased the alpha7 nAChR density in the CA2/3 area and the stratum oriens and increased the non-alpha7 nAChR density in the dentate gyrus.
Conclusion: Tobacco smoke exposure leads to nicotine dependence as indicated by precipitated affective and somatic withdrawal signs and induces an upregulation of nAChRs in the hippocampus.
Figures



Similar articles
-
Role of α7- and β4-containing nicotinic acetylcholine receptors in the affective and somatic aspects of nicotine withdrawal: studies in knockout mice.Behav Genet. 2012 May;42(3):423-36. doi: 10.1007/s10519-011-9511-0. Epub 2011 Oct 19. Behav Genet. 2012. PMID: 22009521 Free PMC article.
-
Preadolescent tobacco smoke exposure leads to acute nicotine dependence but does not affect the rewarding effects of nicotine or nicotine withdrawal in adulthood in rats.Pharmacol Biochem Behav. 2010 Jun;95(4):401-9. doi: 10.1016/j.pbb.2010.02.018. Epub 2010 Mar 6. Pharmacol Biochem Behav. 2010. PMID: 20211642 Free PMC article.
-
Chronic exposure to cigarette smoke extract upregulates nicotinic receptor binding in adult and adolescent rats.Neuropharmacology. 2020 Dec 15;181:108308. doi: 10.1016/j.neuropharm.2020.108308. Epub 2020 Sep 17. Neuropharmacology. 2020. PMID: 32950561 Free PMC article.
-
Subtypes of nicotinic acetylcholine receptors in nicotine reward, dependence, and withdrawal: evidence from genetically modified mice.Behav Pharmacol. 2008 Sep;19(5-6):461-84. doi: 10.1097/FBP.0b013e32830c360e. Behav Pharmacol. 2008. PMID: 18690103 Free PMC article. Review.
-
Sensory Effects of Nicotine and Tobacco.Nicotine Tob Res. 2022 Feb 14;24(3):306-315. doi: 10.1093/ntr/ntab086. Nicotine Tob Res. 2022. PMID: 33955474 Free PMC article. Review.
Cited by
-
Reinforcer devaluation as a consequence of acute nicotine exposure and withdrawal.Psychopharmacology (Berl). 2015 May;232(9):1583-94. doi: 10.1007/s00213-014-3792-y. Epub 2014 Nov 18. Psychopharmacology (Berl). 2015. PMID: 25401169 Free PMC article.
-
Persistent Anhedonia After Intermittent Long-Access Nicotine Self-Administration in Rats.bioRxiv [Preprint]. 2025 Mar 19:2025.03.19.644137. doi: 10.1101/2025.03.19.644137. bioRxiv. 2025. PMID: 40166193 Free PMC article. Preprint.
-
Rodent models for nicotine withdrawal.J Psychopharmacol. 2021 Oct;35(10):1169-1187. doi: 10.1177/02698811211005629. Epub 2021 Apr 22. J Psychopharmacol. 2021. PMID: 33888006 Free PMC article. Review.
-
An Inventory of Methods for the Assessment of Additive Increased Addictiveness of Tobacco Products.Nicotine Tob Res. 2016 Jul;18(7):1546-55. doi: 10.1093/ntr/ntw002. Epub 2016 Jan 26. Nicotine Tob Res. 2016. PMID: 26817491 Free PMC article. Review.
-
Nicotine abolishes memory-related synaptic strengthening and promotes synaptic depression in the neurogenic dentate gyrus of miR-132/212 knockout mice.Addict Biol. 2021 Mar;26(2):e12905. doi: 10.1111/adb.12905. Epub 2020 Apr 15. Addict Biol. 2021. PMID: 32293776 Free PMC article.
References
-
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4. American Psychiatric Press; Washington, DC: 2000. text revision edn.
-
- Anderson KL, Pinkerton KE, Uyeminami D, Simons CT, Carstens MI, Carstens E. Antinociception induced by chronic exposure of rats to cigarette smoke. NeurosciLett. 2004;366:86–91. - PubMed
-
- Aricioglu F, Altunbas H. Harmane induces anxiolysis and antidepressant-like effects in rats. Ann NY Acad Sci. 2003;1009:196–201. - PubMed
-
- Bardo MT, Green TA, Crooks PA, Dwoskin LP. Nornicotine is self-administered intravenously by rats. Psychopharmacology (Berl) 1999;146:290–296. - PubMed
-
- Belluzzi JD, Wang R, Leslie FM. Acetaldehyde enhances acquisition of nicotine self-administration in adolescent rats. Neuropsychopharmacology. 2005;30:705–712. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

