Insights from the crystal structure of the sixth BRCT domain of topoisomerase IIbeta binding protein 1
- PMID: 19937654
- PMCID: PMC2817851
- DOI: 10.1002/pro.290
Insights from the crystal structure of the sixth BRCT domain of topoisomerase IIbeta binding protein 1
Abstract
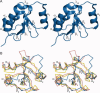
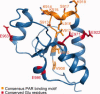
Topoisomerase IIbeta binding protein 1 (TopBP1) is a major player in the DNA damage response and interacts with a number of protein partners via its eight BRCA1 carboxy-terminal (BRCT) domains. In particular, the sixth BRCT domain of TopBP1 has been implicated in binding to the phosphorylated transcription factor, E2F1, and poly(ADP-ribose) polymerase 1 (PARP-1), where the latter interaction is responsible for the poly(ADP-ribosyl)ation of TopBP1. To gain a better understanding of the nature of TopBP1 BRCT6 interactions, we solved the crystal structure of BRCT6 to 1.34 A. The crystal structure reveals a degenerate phospho-peptide binding pocket and lacks conserved hydrophobic residues involved in packing of tandem BRCT repeats, which, together with results from phospho-peptide binding studies, strongly suggest that TopBP1 BRCT6 independently does not function as a phospho-peptide binding domain. We further provide insight into poly(ADP-ribose) binding and sites of potential modification by PARP-1.
Figures

Similar articles
-
The DNA topoisomerase IIbeta binding protein 1 (TopBP1) interacts with poly (ADP-ribose) polymerase (PARP-1).J Cell Biochem. 2007 Sep 1;102(1):171-82. doi: 10.1002/jcb.21292. J Cell Biochem. 2007. PMID: 17340632
-
Crystal structure and mutagenesis of a nucleic acid-binding BRCT domain in human PARP4.J Biol Chem. 2025 Jun;301(6):110277. doi: 10.1016/j.jbc.2025.110277. Epub 2025 May 22. J Biol Chem. 2025. PMID: 40412520 Free PMC article.
-
Structural and biophysical studies of human PARP-1 in complex with damaged DNA.J Mol Biol. 2010 Feb 5;395(5):983-94. doi: 10.1016/j.jmb.2009.11.062. Epub 2009 Dec 4. J Mol Biol. 2010. PMID: 19962992 Free PMC article.
-
Poly(ADP-ribose) polymerase (PARP) inhibitors for the treatment of ovarian cancer.Cochrane Database Syst Rev. 2022 Feb 16;2(2):CD007929. doi: 10.1002/14651858.CD007929.pub4. Cochrane Database Syst Rev. 2022. PMID: 35170751 Free PMC article.
-
Poly-ADP-ribosylation dynamics, signaling, and analysis.Environ Mol Mutagen. 2024 Nov;65(9):315-337. doi: 10.1002/em.22623. Epub 2024 Sep 2. Environ Mol Mutagen. 2024. PMID: 39221603 Review.
Cited by
-
Structural studies of the PARP-1 BRCT domain.BMC Struct Biol. 2011 Oct 3;11:37. doi: 10.1186/1472-6807-11-37. BMC Struct Biol. 2011. PMID: 21967661 Free PMC article.
-
BRCT Domains: Structure, Functions, and Implications in Disease-New Therapeutic Targets for Innovative Drug Discovery against Infections.Pharmaceutics. 2023 Jun 27;15(7):1839. doi: 10.3390/pharmaceutics15071839. Pharmaceutics. 2023. PMID: 37514027 Free PMC article. Review.
-
Phosphopeptide interactions with BRCA1 BRCT domains: More than just a motif.Prog Biophys Mol Biol. 2015 Mar;117(2-3):143-148. doi: 10.1016/j.pbiomolbio.2015.02.003. Epub 2015 Feb 17. Prog Biophys Mol Biol. 2015. PMID: 25701377 Free PMC article. Review.
-
BRCT domains: easy as one, two, three.Cell Cycle. 2011 Aug 1;10(15):2461-70. doi: 10.4161/cc.10.15.16312. Epub 2011 Aug 1. Cell Cycle. 2011. PMID: 21734457 Free PMC article. Review.
-
Structure and function of the Rad9-binding region of the DNA-damage checkpoint adaptor TopBP1.Nucleic Acids Res. 2011 Jan;39(1):313-24. doi: 10.1093/nar/gkq743. Epub 2010 Aug 19. Nucleic Acids Res. 2011. PMID: 20724438 Free PMC article.
References
-
- Garcia V, Furuya K, Carr AM. Identification and functional analysis of TopBP1 and its homologs. DNA Repair (Amst) 2005;4:1227–1239. - PubMed
-
- Kumagai A, Lee J, Yoo HY, Dunphy WG. TopBP1 activates the ATR-ATRIP complex. Cell. 2006;124:943–955. - PubMed
-
- Manke IA, Lowery DM, Nguyen A, Yaffe MB. BRCT repeats as phosphopeptide-binding modules involved in protein targeting. Science. 2003;302:636–639. - PubMed
-
- Yu X, Chini CC, He M, Mer G, Chen J. The BRCT domain is a phospho-protein binding domain. Science. 2003;302:639–632. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

