Characterization of the cytokine and maturation responses of pure populations of porcine plasmacytoid dendritic cells to porcine viruses and toll-like receptor agonists
- PMID: 19939462
- PMCID: PMC7126865
- DOI: 10.1016/j.vetimm.2009.10.026
Characterization of the cytokine and maturation responses of pure populations of porcine plasmacytoid dendritic cells to porcine viruses and toll-like receptor agonists
Abstract
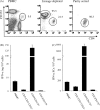
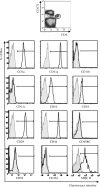
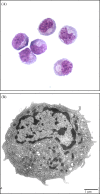
Plausible representatives of plasmacytoid dendritic cells (pDCs) in pigs have been characterized as being CD4(hi)CD172(lo). Due to their paucity in blood, we utilized novel fluorescent-activated cell sorting procedures to isolate them from PBMC. The resultant subset was greater than 98% homogeneous in regards to the selected phenotype and contained the preponderance of individuals secreting IFN-alpha after exposure to a known stimulant, transmissible gastroenteritis virus (TGEV). In addition to being a potent source of IFN-alpha, other properties of these porcine CD4(hi)CD172(lo) cells including their morphological transition from a plasma cell-like shape during quiescence to one resembling a dendritic cell (DC) after activation by TGEV and their relatively strong constitutive expression of interferon regulatory factor-7 (IRF-7) conformed to the expectations of genuine pDCs. While a substantial IFN-alpha response was also elicited from the porcine pDCs by pseudorabies virus (PrV), swine influenza virus (SIV), and TLR7 and 9 agonists, there was an agent-dependent induction of varying amounts of IL-2, IL-6, IL-8, IL-12, IFN-gamma, and TNF-alpha. Notably, porcine reproductive and respiratory syndrome virus (PRRSV) failed to provoke the pDCs to secrete any of the measured cytokines except IL-2. Moreover, whereas pDCs exposed to TGEV or the TLR9 agonist rapidly increased IRF-7 production and morphed into DCs with enhanced CD80/86 expression, similar alterations were not observed during incubation with PRRSV. This atypical response of pDCs to PRRSV may contribute to its pathogenesis, which unlike that associated with PrV, SIV or TGEV includes persistent infection and limited development of protective immunity.
Copyright 2009 Elsevier B.V. All rights reserved.
Figures





Similar articles
-
North American porcine reproductive and respiratory syndrome viruses inhibit type I interferon production by plasmacytoid dendritic cells.J Virol. 2011 Mar;85(6):2703-13. doi: 10.1128/JVI.01616-10. Epub 2010 Dec 29. J Virol. 2011. PMID: 21191013 Free PMC article.
-
Evaluation of the Effect of Inactivated Transmissible Gastroenteritis Virus Vaccine with Nano Silicon on the Phenotype and Function of Porcine Dendritic Cells.Viruses. 2021 Oct 26;13(11):2158. doi: 10.3390/v13112158. Viruses. 2021. PMID: 34834964 Free PMC article.
-
CD163 and pAPN double-knockout pigs are resistant to PRRSV and TGEV and exhibit decreased susceptibility to PDCoV while maintaining normal production performance.Elife. 2020 Sep 2;9:e57132. doi: 10.7554/eLife.57132. Elife. 2020. PMID: 32876563 Free PMC article.
-
Porcine innate and adaptative immune responses to influenza and coronavirus infections.Ann N Y Acad Sci. 2006 Oct;1081(1):130-6. doi: 10.1196/annals.1373.014. Ann N Y Acad Sci. 2006. PMID: 17135502 Free PMC article. Review.
-
Swine Dendritic Cell Response to Porcine Reproductive and Respiratory Syndrome Virus: An Update.Front Immunol. 2021 Jul 28;12:712109. doi: 10.3389/fimmu.2021.712109. eCollection 2021. Front Immunol. 2021. PMID: 34394113 Free PMC article. Review.
Cited by
-
A genome-wide association study of fetal response to type 2 porcine reproductive and respiratory syndrome virus challenge.Sci Rep. 2016 Feb 5;6:20305. doi: 10.1038/srep20305. Sci Rep. 2016. PMID: 26846722 Free PMC article.
-
Porcine Plasmacytoid Dendritic Cells Are Unique in Their Expression of a Functional NKp46 Receptor.Front Immunol. 2022 Mar 11;13:822258. doi: 10.3389/fimmu.2022.822258. eCollection 2022. Front Immunol. 2022. PMID: 35371050 Free PMC article.
-
Regulation of porcine plasmacytoid dendritic cells by cytokines.PLoS One. 2013;8(4):e60893. doi: 10.1371/journal.pone.0060893. Epub 2013 Apr 8. PLoS One. 2013. PMID: 23577175 Free PMC article.
-
Cellular Innate Immunity against PRRSV and Swine Influenza Viruses.Vet Sci. 2019 Mar 11;6(1):26. doi: 10.3390/vetsci6010026. Vet Sci. 2019. PMID: 30862035 Free PMC article. Review.
-
North American porcine reproductive and respiratory syndrome viruses inhibit type I interferon production by plasmacytoid dendritic cells.J Virol. 2011 Mar;85(6):2703-13. doi: 10.1128/JVI.01616-10. Epub 2010 Dec 29. J Virol. 2011. PMID: 21191013 Free PMC article.
References
-
- Albina E., Carrat C., Charley B. Interferon-alpha response to swine arterivirus (PoAV), the porcine reproductive and respiratory syndrome virus. J. Interferon Cytokine Res. 1998;18:485–490. - PubMed
-
- Alvarez B., Domenech N., Alonso F., Sanchez C., Gomez del Moral M., Ezquerra A., Dominguez J. Molecular and functional characterization of porcine LFA-1 using monoclonal antibodies to CD11a and CD18. Xenotransplantation. 2000;7:258–266. - PubMed
-
- Asselin-Paturel C., Boonstra A., Dalod M., Durand I., Yessaad N., Dezutter-Dambuyant C., Vicari A., O’Garra A., Biron C., Briere F., Trinchieri G. Mouse type I IFN-producing cells are immature APCs with plasmacytoid morphology. Nat. Immunol. 2001;2:1144–1150. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

