Lipid bilayer regulation of membrane protein function: gramicidin channels as molecular force probes
- PMID: 19940001
- PMCID: PMC2842803
- DOI: 10.1098/rsif.2009.0443
Lipid bilayer regulation of membrane protein function: gramicidin channels as molecular force probes
Abstract
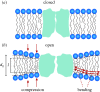
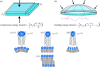
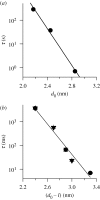
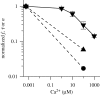
Membrane protein function is regulated by the host lipid bilayer composition. This regulation may depend on specific chemical interactions between proteins and individual molecules in the bilayer, as well as on non-specific interactions between proteins and the bilayer behaving as a physical entity with collective physical properties (e.g. thickness, intrinsic monolayer curvature or elastic moduli). Studies in physico-chemical model systems have demonstrated that changes in bilayer physical properties can regulate membrane protein function by altering the energetic cost of the bilayer deformation associated with a protein conformational change. This type of regulation is well characterized, and its mechanistic elucidation is an interdisciplinary field bordering on physics, chemistry and biology. Changes in lipid composition that alter bilayer physical properties (including cholesterol, polyunsaturated fatty acids, other lipid metabolites and amphiphiles) regulate a wide range of membrane proteins in a seemingly non-specific manner. The commonality of the changes in protein function suggests an underlying physical mechanism, and recent studies show that at least some of the changes are caused by altered bilayer physical properties. This advance is because of the introduction of new tools for studying lipid bilayer regulation of protein function. The present review provides an introduction to the regulation of membrane protein function by the bilayer physical properties. We further describe the use of gramicidin channels as molecular force probes for studying this mechanism, with a unique ability to discriminate between consequences of changes in monolayer curvature and bilayer elastic moduli.
Figures




Similar articles
-
Amphiphile regulation of ion channel function by changes in the bilayer spring constant.Proc Natl Acad Sci U S A. 2010 Aug 31;107(35):15427-30. doi: 10.1073/pnas.1007455107. Epub 2010 Aug 16. Proc Natl Acad Sci U S A. 2010. PMID: 20713738 Free PMC article.
-
Regulation of sodium channel function by bilayer elasticity: the importance of hydrophobic coupling. Effects of Micelle-forming amphiphiles and cholesterol.J Gen Physiol. 2004 May;123(5):599-621. doi: 10.1085/jgp.200308996. J Gen Physiol. 2004. PMID: 15111647 Free PMC article.
-
Single-molecule methods for monitoring changes in bilayer elastic properties.Methods Mol Biol. 2007;400:543-70. doi: 10.1007/978-1-59745-519-0_37. Methods Mol Biol. 2007. PMID: 17951759 Review.
-
Bilayer thickness and membrane protein function: an energetic perspective.Annu Rev Biophys Biomol Struct. 2007;36:107-30. doi: 10.1146/annurev.biophys.36.040306.132643. Annu Rev Biophys Biomol Struct. 2007. PMID: 17263662 Review.
-
A one-dimensional continuum elastic model for membrane-embedded gramicidin dimer dissociation.PLoS One. 2011 Feb 4;6(2):e15563. doi: 10.1371/journal.pone.0015563. PLoS One. 2011. PMID: 21326605 Free PMC article.
Cited by
-
Activation of the Human Epithelial Sodium Channel (ENaC) by Bile Acids Involves the Degenerin Site.J Biol Chem. 2016 Sep 16;291(38):19835-47. doi: 10.1074/jbc.M116.726471. Epub 2016 Aug 3. J Biol Chem. 2016. PMID: 27489102 Free PMC article.
-
Regulation of Antimicrobial Peptide Activity via Tuning Deformation Fields by Membrane-Deforming Inclusions.Int J Mol Sci. 2021 Dec 28;23(1):326. doi: 10.3390/ijms23010326. Int J Mol Sci. 2021. PMID: 35008752 Free PMC article.
-
Divergent effects of anesthetics on lipid bilayer properties and sodium channel function.Eur Biophys J. 2017 Oct;46(7):617-626. doi: 10.1007/s00249-017-1239-1. Epub 2017 Jul 10. Eur Biophys J. 2017. PMID: 28695248 Free PMC article. Review.
-
β1-Integrin-Mediated Adhesion Is Lipid-Bilayer Dependent.Biophys J. 2017 Sep 5;113(5):1080-1092. doi: 10.1016/j.bpj.2017.07.010. Biophys J. 2017. PMID: 28877491 Free PMC article.
-
Membrane-Mediated Lateral Interactions Regulate the Lifetime of Gramicidin Channels.Membranes (Basel). 2020 Nov 25;10(12):368. doi: 10.3390/membranes10120368. Membranes (Basel). 2020. PMID: 33255806 Free PMC article.
References
-
- Adachi S., Nagao T., Ingólfsson H. I., Maxfield F. R., Andersen O. S., Kopelovich L., Weinstein I. B. 2007. The inhibitory effect of (-)-epigallocatechin gallate on activation of the epidermal growth factor receptor is associated with altered lipid order in HT29 colon cancer cells. Cancer Res. 67, 6493–6501. (10.1158/0008-5472.CAN-07-0411) - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

