Fentanyl buccal soluble film (FBSF) for breakthrough pain in patients with cancer: a randomized, double-blind, placebo-controlled study
- PMID: 19940014
- PMCID: PMC2875549
- DOI: 10.1093/annonc/mdp541
Fentanyl buccal soluble film (FBSF) for breakthrough pain in patients with cancer: a randomized, double-blind, placebo-controlled study
Abstract
Background: Fentanyl buccal soluble film (FBSF) has been developed as a treatment of breakthrough pain in opioid-tolerant patients with cancer. The objective of this study was to evaluate the efficacy of FBSF at doses of 200-1200 microg in the management of breakthrough pain in patients with cancer receiving ongoing opioid therapy.
Patients and methods: This was a multicenter, randomized, double-blind, placebo-controlled, multiple-crossover study that included opioid-tolerant adult patients with chronic cancer pain who experienced one to four daily episodes of breakthrough pain. The primary efficacy assessment was the sum of pain intensity differences at 30 min (SPID30) postdose.
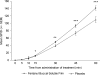
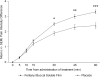
Results: The intent-to-treat population consisted of 80 patients with > or =1 post-baseline efficacy assessment. The least-squares mean (LSM +/- SEM) of the SPID30 was significantly greater for FBSF-treated episodes of breakthrough pain than for placebo-treated episodes (47.9 +/- 3.9 versus 38.1 +/- 4.3; P = 0.004). There was statistical separation from placebo starting at 15 min up through 60 min (last time point assessed). There were no unexpected adverse events (AEs) or clinically significant safety findings.
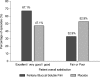
Conclusions: FBSF is an effective option for control of breakthrough pain in patients receiving ongoing opioid therapy. In this study, FBSF was well tolerated in the oral cavity, with no reports of treatment-related oral AEs.
Figures
Similar articles
-
Fentanyl Buccal Soluble Film: A Review in Breakthrough Cancer Pain.Clin Drug Investig. 2016 May;36(5):413-9. doi: 10.1007/s40261-016-0394-y. Clin Drug Investig. 2016. PMID: 27007271 Review.
-
Fentanyl buccal tablet for the relief of breakthrough pain in opioid-tolerant adult patients with chronic neuropathic pain: a multicenter, randomized, double-blind, placebo-controlled study.Clin Ther. 2007 Apr;29(4):588-601. doi: 10.1016/j.clinthera.2007.04.007. Clin Ther. 2007. PMID: 17617282 Clinical Trial.
-
Efficacy and tolerability of intranasal fentanyl spray 50 to 200 microg for breakthrough pain in patients with cancer: a phase III, multinational, randomized, double-blind, placebo-controlled, crossover trial with a 10-month, open-label extension treatment period.Clin Ther. 2009 Jun;31(6):1177-91. doi: 10.1016/j.clinthera.2009.05.022. Clin Ther. 2009. PMID: 19695386 Clinical Trial.
-
A dose titration study of fentanyl buccal soluble film for breakthrough cancer pain in Taiwan.Cancer Rep (Hoboken). 2019 Oct;2(5):e1179. doi: 10.1002/cnr2.1179. Epub 2019 Apr 23. Cancer Rep (Hoboken). 2019. PMID: 32721110 Free PMC article. Clinical Trial.
-
Fentanyl buccal tablet.Drugs Today (Barc). 2008 Jan;44(1):41-54. doi: 10.1358/dot.2008.44.1.1178469. Drugs Today (Barc). 2008. PMID: 18301803 Review.
Cited by
-
Pharmacotherapy for breakthrough cancer pain.Drugs. 2012 Jan 22;72(2):181-90. doi: 10.2165/11597260-000000000-00000. Drugs. 2012. PMID: 22233484 Review.
-
Fentanyl buccal tablet for breakthrough cancer pain in clinical practice: results of the non-interventional prospective study ErkentNIS.Support Care Cancer. 2018 Feb;26(2):491-497. doi: 10.1007/s00520-017-3853-y. Epub 2017 Aug 28. Support Care Cancer. 2018. PMID: 28849261 Free PMC article.
-
Abstral (Fentanyl Sublingual Tablets for Breakthrough Cancer Pain).P T. 2011 Feb;36(2):2-28. P T. 2011. PMID: 21560267 Free PMC article. No abstract available.
-
Breakthrough cancer pain (BTcP): a synthesis of taxonomy, pathogenesis, therapy, and good clinical practice in adult patients in Italy.Adv Ther. 2014 Jul;31(7):657-82. doi: 10.1007/s12325-014-0130-z. Epub 2014 Jul 9. Adv Ther. 2014. PMID: 25005168 Free PMC article. Review.
-
Fentanyl Buccal Soluble Film: A Review in Breakthrough Cancer Pain.Clin Drug Investig. 2016 May;36(5):413-9. doi: 10.1007/s40261-016-0394-y. Clin Drug Investig. 2016. PMID: 27007271 Review.
References
-
- Portenoy RK, Hagen NA. Breakthrough pain: definition, prevalence and characteristics. Pain. 1990;41:273–281. - PubMed
-
- Caraceni A, Martini C, Zecca E, et al. Breakthrough pain characteristics and syndromes in patients with cancer pain. An international survey. Palliat Med. 2004;18:177–183. - PubMed
-
- Farrar JT, Cleary J, Rauck R, et al. Oral transmucosal fentanyl citrate: randomized, double-blinded, placebo-controlled trial for treatment of breakthrough pain in cancer patients. J Natl Cancer Inst. 1998;90:611–616. - PubMed
-
- William L, Macleod R. Management of breakthrough pain in patients with cancer. Drugs. 2008;68:913–924. - PubMed
-
- Breivik H, Cherny N, Collett B, et al. Cancer-related pain: a pan-European survey of prevalence, treatment, and patient attitudes. Ann Oncol. 2009;20:1420–1433. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

