Constitutive activity of the human TRPML2 channel induces cell degeneration
- PMID: 19940139
- PMCID: PMC2807332
- DOI: 10.1074/jbc.M109.046508
Constitutive activity of the human TRPML2 channel induces cell degeneration
Abstract
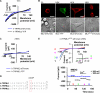
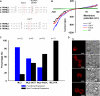
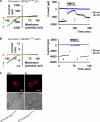
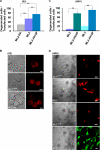
The mucolipin (TRPML) ion channel proteins represent a distinct subfamily of channel proteins within the transient receptor potential (TRP) superfamily of cation channels. Mucolipin 1, 2, and 3 (TRPML1, -2, and -3, respectively) are channel proteins that share high sequence homology with each other and homology in the transmembrane domain with other TRPs. Mutations in the TRPML1 protein are implicated in mucolipidosis type IV, whereas mutations in TRPML3 are found in the varitint-waddler mouse. The properties of the wild type TRPML2 channel are not well known. Here we show functional expression of the wild type human TRPML2 channel (h-TRPML2). The channel is functional at the plasma membrane and characterized by a significant inward rectification similar to other constitutively active TRPML mutant isoforms. The h-TRPML2 channel displays nonselective cation permeability, which is Ca(2+)-permeable and inhibited by low extracytosolic pH but not Ca(2+) regulated. In addition, constitutively active h-TRPML2 leads to cell death by causing Ca(2+) overload. Furthermore, we demonstrate by functional mutation analysis that h-TRPML2 shares similar characteristics and structural similarities with other TRPML channels that regulate the channel in a similar manner. Hence, in addition to overall structure, all three TRPML channels also share common modes of regulation.
Figures


Similar articles
-
Drosophila TRPML forms PI(3,5)P2-activated cation channels in both endolysosomes and plasma membrane.J Biol Chem. 2014 Feb 14;289(7):4262-72. doi: 10.1074/jbc.M113.506501. Epub 2013 Dec 27. J Biol Chem. 2014. PMID: 24375408 Free PMC article.
-
Functional multimerization of mucolipin channel proteins.J Cell Physiol. 2010 Feb;222(2):328-35. doi: 10.1002/jcp.21956. J Cell Physiol. 2010. PMID: 19885840
-
The tissue-specific expression of TRPML2 (MCOLN-2) gene is influenced by the presence of TRPML1.Pflugers Arch. 2009 Nov;459(1):79-91. doi: 10.1007/s00424-009-0716-5. Pflugers Arch. 2009. PMID: 19763610 Free PMC article.
-
TRPML2 and mucolipin evolution.Handb Exp Pharmacol. 2014;222:647-58. doi: 10.1007/978-3-642-54215-2_25. Handb Exp Pharmacol. 2014. PMID: 24756724 Review.
-
The mucolipin-2 (TRPML2) ion channel: a tissue-specific protein crucial to normal cell function.Pflugers Arch. 2016 Feb;468(2):177-92. doi: 10.1007/s00424-015-1732-2. Epub 2015 Sep 4. Pflugers Arch. 2016. PMID: 26336837 Free PMC article. Review.
Cited by
-
Ion channels in cancer: future perspectives and clinical potential.Philos Trans R Soc Lond B Biol Sci. 2014 Feb 3;369(1638):20130108. doi: 10.1098/rstb.2013.0108. Print 2014 Mar 19. Philos Trans R Soc Lond B Biol Sci. 2014. PMID: 24493756 Free PMC article. Review.
-
Overexpression of transient receptor potential mucolipin-2 ion channels in gliomas: role in tumor growth and progression.Oncotarget. 2016 Jul 12;7(28):43654-43668. doi: 10.18632/oncotarget.9661. Oncotarget. 2016. PMID: 27248469 Free PMC article.
-
TNF inhibitors target a mevalonate metabolite/TRPM2/calcium signaling axis in neutrophils to dampen vasculitis in Behçet's disease.Nat Commun. 2024 Oct 26;15(1):9261. doi: 10.1038/s41467-024-53528-3. Nat Commun. 2024. PMID: 39461948 Free PMC article.
-
Properties of the TRPML3 channel pore and its stable expansion by the Varitint-Waddler-causing mutation.J Biol Chem. 2010 May 28;285(22):16513-20. doi: 10.1074/jbc.M109.078204. Epub 2010 Apr 8. J Biol Chem. 2010. PMID: 20378547 Free PMC article.
-
Opening the TRPML gates.Chem Biol. 2010 Mar 26;17(3):209-10. doi: 10.1016/j.chembiol.2010.02.009. Chem Biol. 2010. PMID: 20338511 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

