Peptidyl-prolyl isomerase 1 regulates protein phosphatase 2A-mediated topographic phosphorylation of neurofilament proteins
- PMID: 19940183
- PMCID: PMC3849796
- DOI: 10.1523/JNEUROSCI.4469-09.2009
Peptidyl-prolyl isomerase 1 regulates protein phosphatase 2A-mediated topographic phosphorylation of neurofilament proteins
Abstract
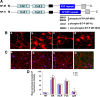
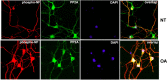
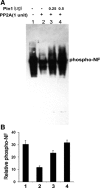
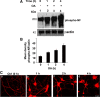
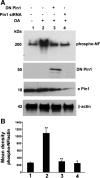
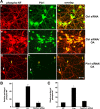
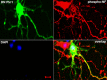
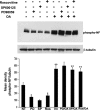
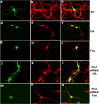
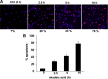
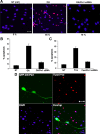
In normal neurons, neurofilament (NF) proteins are phosphorylated in the axonal compartment. However, in neurodegenerative disorders such as Alzheimer's disease (AD), Parkinson's disease (PD), and amyotrophic lateral sclerosis (ALS), NF proteins are aberrantly hyperphosphorylated within the cell bodies. The aberrant hyperphosphorylation of NF accumulations found in neurodegeneration could be attributable to either deregulation of proline-directed Ser/Thr kinase(s) activity or downregulation of protein phosphatase(s) activity. In this study, we found that protein phosphatase 2A (PP2A) expression is high in neuronal cell bodies and that inhibition of PP2A activity by okadaic acid (OA), microcystin LR (mLR), or fostriecin (Fos) leads to perikaryal hyperphosphorylation of NF. Peptidyl-prolyl isomerase Pin1 inhibits the dephosphorylation of NF by PP2A in vitro. In cortical neurons, Pin1 modulates the topographic phosphorylation of the proline-directed Ser/Thr residues within the tail domain of NF proteins by inhibiting the dephosphorylation by PP2A. Inhibition of Pin1 inhibits OA-induced aberrant perikaryal phosphorylation of NF. Treatment of cortical neurons with OA or Fos prevents the general anterograde transport of transfected green fluorescent protein-high-molecular-mass (NF-H) into axons caused by hyperphosphorylation of NF-H, and inhibition of Pin1 rescues this effect. Furthermore, inhibition of Pin1 inhibits the OA- or Fos-induced neuronal apoptosis. We show that OA-induced hyperphosphorylation of NF is a consequence of dephosphorylation of NF and is independent of c-Jun N-terminal protein kinase, extracellular signal-regulated kinase, and cyclin-dependent kinase-5 pathways. This study highlights a novel signaling role of PP2A by Pin1 and implicates Pin1 as a therapeutic target to reduce aberrant phosphorylation of NF proteins in neurodegenerative disorders such as AD, PD, and ALS.
Figures
Similar articles
-
Pin1-dependent prolyl isomerization modulates the stress-induced phosphorylation of high molecular weight neurofilament protein.J Biol Chem. 2008 Sep 26;283(39):26737-47. doi: 10.1074/jbc.M801633200. Epub 2008 Jul 17. J Biol Chem. 2008. PMID: 18635547 Free PMC article.
-
Inhibition of Pin1 reduces glutamate-induced perikaryal accumulation of phosphorylated neurofilament-H in neurons.Mol Biol Cell. 2007 Sep;18(9):3645-55. doi: 10.1091/mbc.e07-03-0237. Epub 2007 Jul 11. Mol Biol Cell. 2007. PMID: 17626162 Free PMC article.
-
Phosphorylation-specific peptidyl-prolyl isomerization of neuronal cytoskeletal proteins by Pin1: implications for therapeutics in neurodegeneration.J Alzheimers Dis. 2010;19(2):389-403. doi: 10.3233/JAD-2010-1243. J Alzheimers Dis. 2010. PMID: 20110589 Review.
-
Topographic regulation of neuronal intermediate filaments by phosphorylation, role of peptidyl-prolyl isomerase 1: significance in neurodegeneration.Histochem Cell Biol. 2013 Jul;140(1):23-32. doi: 10.1007/s00418-013-1108-7. Epub 2013 Jun 23. Histochem Cell Biol. 2013. PMID: 23793952 Review.
-
Isomerase Pin1 stimulates dephosphorylation of tau protein at cyclin-dependent kinase (Cdk5)-dependent Alzheimer phosphorylation sites.J Biol Chem. 2013 Mar 15;288(11):7968-7977. doi: 10.1074/jbc.M112.433326. Epub 2013 Jan 28. J Biol Chem. 2013. PMID: 23362255 Free PMC article.
Cited by
-
Prolyl isomerase Pin1 regulates neuronal differentiation via β-catenin.Mol Cell Biol. 2012 Aug;32(15):2966-78. doi: 10.1128/MCB.05688-11. Epub 2012 May 29. Mol Cell Biol. 2012. PMID: 22645310 Free PMC article.
-
Use of okadaic acid to identify relevant phosphoepitopes in pathology: a focus on neurodegeneration.Mar Drugs. 2013 May 21;11(5):1656-68. doi: 10.3390/md11051656. Mar Drugs. 2013. PMID: 23697949 Free PMC article. Review.
-
Quantitative phosphoproteomic analysis of neuronal intermediate filament proteins (NF-M/H) in Alzheimer's disease by iTRAQ.FASEB J. 2010 Nov;24(11):4396-407. doi: 10.1096/fj.10-157859. Epub 2010 Jul 12. FASEB J. 2010. PMID: 20624930 Free PMC article.
-
Prolyl isomerase Pin1 as a molecular switch to determine the fate of phosphoproteins.Trends Biochem Sci. 2011 Oct;36(10):501-14. doi: 10.1016/j.tibs.2011.07.001. Epub 2011 Aug 17. Trends Biochem Sci. 2011. PMID: 21852138 Free PMC article. Review.
-
Neuroanatomical Quantitative Proteomics Reveals Common Pathogenic Biological Routes between Amyotrophic Lateral Sclerosis (ALS) and Frontotemporal Dementia (FTD).Int J Mol Sci. 2018 Dec 20;20(1):4. doi: 10.3390/ijms20010004. Int J Mol Sci. 2018. PMID: 30577465 Free PMC article. Review.
References
-
- Ahlijanian MK, Barrezueta NX, Williams RD, Jakowski A, Kowsz KP, McCarthy S, Coskran T, Carlo A, Seymour PA, Burkhardt JE, Nelson RB, McNeish JD. Hyperphosphorylated tau and neurofilament and cytoskeletal disruptions in mice overexpressing human p25, an activator of cdk5. Proc Natl Acad Sci U S A. 2000;97:2910–2915. - PMC - PubMed
-
- Dashiell SM, Tanner SL, Pant HC, Quarles RH. Myelin-associated glycoprotein modulates expression and phosphorylation of neuronal cytoskeletal elements and their associated kinases. J Neurochem. 2002;81:1263–1272. - PubMed
-
- de Waegh SM, Lee VM, Brady ST. Local modulation of neurofilament phosphorylation, axonal caliber and slow axonal transport by mylenating Schwann cells. Cell. 1992;68:451–463. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous
