Distinct muscarinic acetylcholine receptor subtypes contribute to stability and growth, but not compensatory plasticity, of neuromuscular synapses
- PMID: 19940190
- PMCID: PMC2822401
- DOI: 10.1523/JNEUROSCI.2276-09.2009
Distinct muscarinic acetylcholine receptor subtypes contribute to stability and growth, but not compensatory plasticity, of neuromuscular synapses
Abstract
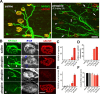
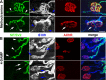
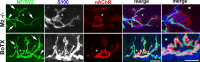
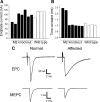
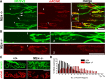
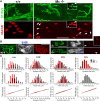
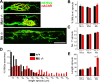
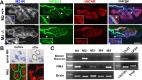
Muscarinic acetylcholine receptors (mAChRs) modulate synaptic function, but whether they influence synaptic structure remains unknown. At neuromuscular junctions (NMJs), mAChRs have been implicated in compensatory sprouting of axon terminals in paralyzed or denervated muscles. Here we used pharmacological and genetic inhibition and localization studies of mAChR subtypes at mouse NMJs to demonstrate their roles in synaptic stability and growth but not in compensatory sprouting. M(2) mAChRs were present solely in motor neurons, whereas M(1), M(3), and M(5) mAChRs were associated with Schwann cells and/or muscle fibers. Blockade of all five mAChR subtypes with atropine evoked pronounced effects, including terminal sprouting, terminal withdrawal, and muscle fiber atrophy. In contrast, methoctramine, an M(2/4)-preferring antagonist, induced terminal sprouting and terminal withdrawal, but no muscle fiber atrophy. Consistent with this observation, M(2)(-/-) but no other mAChR mutant mice exhibited spontaneous sprouting accompanied by extensive loss of parental terminal arbors. Terminal sprouting, however, seemed not to be the causative defect because partial loss of terminal branches was common even in the M(2)(-/-) NMJs without sprouting. Moreover, compensatory sprouting after paralysis or partial denervation was normal in mice deficient in M(2) or other mAChR subtypes. We also found that many NMJs of M(5)(-/-) mice were exceptionally small and reduced in proportion to the size of parental muscle fibers. These findings show that axon terminals are unstable without M(2) and that muscle fiber growth is defective without M(5). Subtype-specific muscarinic signaling provides a novel means for coordinating activity-dependent development and maintenance of the tripartite synapse.
Figures

Similar articles
-
Ciliary neurotrophic factor is not required for terminal sprouting and compensatory reinnervation of neuromuscular synapses: re-evaluation of CNTF null mice.Exp Neurol. 2007 Jun;205(2):437-48. doi: 10.1016/j.expneurol.2007.03.011. Epub 2007 Mar 21. Exp Neurol. 2007. PMID: 17445802 Free PMC article.
-
The expression of the chemorepellent Semaphorin 3A is selectively induced in terminal Schwann cells of a subset of neuromuscular synapses that display limited anatomical plasticity and enhanced vulnerability in motor neuron disease.Mol Cell Neurosci. 2006 May-Jun;32(1-2):102-17. doi: 10.1016/j.mcn.2006.03.002. Epub 2006 May 3. Mol Cell Neurosci. 2006. PMID: 16677822
-
Properties of Glial Cell at the Neuromuscular Junction Are Incompatible with Synaptic Repair in the SOD1G37R ALS Mouse Model.J Neurosci. 2020 Sep 30;40(40):7759-7777. doi: 10.1523/JNEUROSCI.1748-18.2020. Epub 2020 Aug 28. J Neurosci. 2020. PMID: 32859714 Free PMC article.
-
Perisynaptic schwann cells - The multitasking cells at the developing neuromuscular junctions.Semin Cell Dev Biol. 2020 Aug;104:31-38. doi: 10.1016/j.semcdb.2020.02.011. Epub 2020 Mar 5. Semin Cell Dev Biol. 2020. PMID: 32147379 Review.
-
Glutamate at the Vertebrate Neuromuscular Junction: From Modulation to Neurotransmission.Cells. 2019 Aug 28;8(9):996. doi: 10.3390/cells8090996. Cells. 2019. PMID: 31466388 Free PMC article. Review.
Cited by
-
The mechanism of choline-mediated inhibition of acetylcholine release in mouse motor synapses.Acta Naturae. 2014 Oct;6(4):110-5. Acta Naturae. 2014. PMID: 25558401 Free PMC article.
-
Early and persistent abnormal decoding by glial cells at the neuromuscular junction in an ALS model.J Neurosci. 2015 Jan 14;35(2):688-706. doi: 10.1523/JNEUROSCI.1379-14.2015. J Neurosci. 2015. PMID: 25589763 Free PMC article.
-
A multilevel screening pipeline in zebrafish identifies therapeutic drugs for GAN.EMBO Mol Med. 2023 Jul 10;15(7):e16267. doi: 10.15252/emmm.202216267. Epub 2023 May 5. EMBO Mol Med. 2023. PMID: 37144692 Free PMC article.
-
Functional adaptation of glial cells at neuromuscular junctions in response to injury.Glia. 2022 Sep;70(9):1605-1629. doi: 10.1002/glia.24184. Epub 2022 Apr 27. Glia. 2022. PMID: 35474470 Free PMC article.
-
The interaction between tropomyosin-related kinase B receptors and presynaptic muscarinic receptors modulates transmitter release in adult rodent motor nerve terminals.J Neurosci. 2010 Dec 8;30(49):16514-22. doi: 10.1523/JNEUROSCI.2676-10.2010. J Neurosci. 2010. PMID: 21147991 Free PMC article.
References
-
- Albani M, Lowrie MB, Vrbová G. Reorganization of motor units in reinnervated muscles of the rat. J Neurol Sci. 1988;88:195–206. - PubMed
-
- Angaut-Petit D, Molgó J, Faille L, Juzans P, Takahashi M. Incorporation of synaptotagmin II to the axolemma of botulinum type-A poisoned mouse motor endings during enhanced quantal acetylcholine release. Brain Res. 1998;797:357–360. - PubMed
-
- Argentieri TM, Aiken SP, Laxminarayan S, McArdle JJ. Characteristics of synaptic transmission in reinnervating rat skeletal muscle. Pflugers Arch. 1992;421:256–261. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
