Lifestyle modification and metformin as long-term treatment options for obese adolescents: study protocol
- PMID: 19943971
- PMCID: PMC2797799
- DOI: 10.1186/1471-2458-9-434
Lifestyle modification and metformin as long-term treatment options for obese adolescents: study protocol
Abstract
Background: Childhood obesity is a serious health concern affecting over 155 million children in developed countries worldwide. Childhood obesity is associated with significantly increased risk for development of type 2 diabetes, cardiovascular disease and psychosocial functioning problems (i.e., depression and decreased quality of life). The two major strategies for management of obesity and associated metabolic abnormalities are lifestyle modification and pharmacologic therapy. This paper will provide the background rationale and methods of the REACH childhood obesity treatment program.
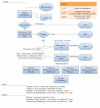
Methods/design: The REACH study is a 2-year multidisciplinary, family-based, childhood obesity treatment program. Seventy-two obese adolescents (aged 10-16 years) and their parents are being recruited to participate in this randomized placebo controlled trial. Participants are randomized to receive either metformin or placebo, and are then randomized to a moderate or a vigorous intensity supervised exercise program for the first 12-weeks. After the 12-week exercise program, participants engage in weekly exercise sessions with an exercise facilitator at a local community center. Participants engage in treatment sessions with a dietitian and social worker monthly for the first year, and then every three months for the second year. The primary outcome measure is change in body mass index and the secondary outcome measures are changes in body composition, risk factors for type 2 diabetes and cardiovascular disease, changes in diet, physical activity, and psychosocial well-being (e.g., quality of life). It is hypothesized that participants who take metformin and engage in vigorous intensity exercise will show the greatest improvements in body mass index. In addition, it is hypothesized that participants who adhere to the REACH program will show improvements in body composition, physical activity, diet, psychosocial functioning and risk factor profiles for type 2 diabetes and cardiovascular disease. These improvements are expected to be maintained over the 2-year program.
Discussion: The findings from this study will advance the knowledge regarding the long-term efficacy and sustainability of interventions for childhood obesity.
Trial registration: ClinicalTrials.gov number NCT00934570.
References
-
- The global epidemic. http://www.iotf.org/intro/global.htm
-
- Merrifield R. Healthy weights for healthy kids: report of the standing committee on health. Ottawa. 2007.