Moderate recurrent hypoglycemia during early development leads to persistent changes in affective behavior in the rat
- PMID: 19944751
- PMCID: PMC2885529
- DOI: 10.1016/j.bbi.2009.11.013
Moderate recurrent hypoglycemia during early development leads to persistent changes in affective behavior in the rat
Abstract
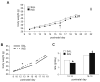
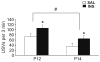
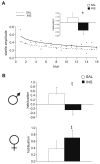
Recurrent hypoglycemia is a common problem among infants and children that is associated with several metabolic disorders and insulin-dependent diabetes mellitus. Although studies have reported a relationship between a history of juvenile hypoglycemia and psychological health problems, the direct effects of recurrent moderate hypoglycemia have not been fully determined. Thus, in this study, we used an animal model to examine the effects of recurrent hypoglycemia during the juvenile period on affective, social, and motor function (assessed under euglycemic conditions) across development. To model recurrent hypoglycemia, rats were administered 5 U/kg of insulin or saline twice per day from postnatal day (P)10 to P19. Body weight gain was retarded in insulin-treated rats during the treatment period, but recovered by the end of treatment. However, insulin-treated rats displayed increases in affective reactivity that emerged early during treatment and persisted after treatment into early adulthood. Specifically, insulin-treated pups showed increased maternal separation-induced vocalizations as infants, and an exaggerated acoustic startle reflex as juveniles and young adults. Moreover, young adult rats with a history of recurrent juvenile hypoglycemia exhibited increased fear-potentiated startle and increases in behavioral and hormonal responses to restraint stress. Some of these effects were sex-dependent. The changes in affective behavior in insulin-exposed pups were accompanied by decreases in adolescent social play behavior. These results provide evidence that recurrent, transient hypoglycemia during juvenile development can lead to increases in fear-related behavior and stress reactivity. Importantly, these phenotypes are not reversed with normalization of blood glucose and may persist into adulthood.
Copyright 2009 Elsevier Inc. All rights reserved.
Figures



References
-
- Akyol A, Kiylioglu N, Bolukbasi O, Guney E, Yurekli Y. Repeated hypoglycemia and cognitive decline. A case report Neuroendocrinol Lett. 2003;24:54–56. - PubMed
-
- Arai M, Widmaier EP. Activation of the pituitary-adrenocortical axis in day-old rats by insulin-induced hypoglycemia. Endocrinology. 1991;129:1505–1512. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

