Structure of a two-G-tetrad intramolecular G-quadruplex formed by a variant human telomeric sequence in K+ solution: insights into the interconversion of human telomeric G-quadruplex structures
- PMID: 19946019
- PMCID: PMC2817458
- DOI: 10.1093/nar/gkp1029
Structure of a two-G-tetrad intramolecular G-quadruplex formed by a variant human telomeric sequence in K+ solution: insights into the interconversion of human telomeric G-quadruplex structures
Abstract
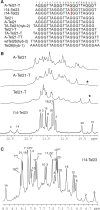
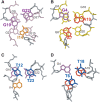
Human telomeric DNA G-quadruplex has been considered as an attractive target for cancer therapeutic intervention. The telomeric sequence shows intrinsic structure polymorphism. Here we report a novel intramolecular G-quadruplex structure formed by a variant human telomeric sequence in K(+) solution. This sequence forms a basket-type intramolecular G-quadruplex with only two G-tetrads but multiple-layer capping structures formed by loop residues. While it is shown that this structure can only be detected in the specifically truncated telomeric sequences without any 5'-flanking residues, our results suggest that this two-G-tetrad conformation is likely to be an intermediate form of the interconversion of different telomeric G-quadruplex conformations.
Figures






References
-
- Blackburn EH. Telomere states and cell fates. Nature. 2000;408:53–56. - PubMed
-
- van Steensel B, Smogorzewska A, de Lange T. TRF2 protects human telomeres from end-to-end fusions. Cell. 1998;92:401–413. - PubMed
-
- Hackett JA, Feldser DM, Greider CW. Telomere dysfunction increases mutation rate and genomic instability. Cell. 2001;106:275–286. - PubMed
-
- Mergny JL, Helene C. G-quadruplex DNA: A target for drug design. Nat. Med. 1998;4:1366–1367. - PubMed
-
- Sun DY, Hurley LH. Targeting telomeres and telomerase. In: Chaires JB, Waring MJ, editors. Methods in Enzymology, Drug-Nucleic Acid Interactions. Vol. 340. San Diego: Academic Press Inc; 2001. pp. 573–592. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

