Identification and expression analysis of ras gene in silkworm, Bombyx mori
- PMID: 19946625
- PMCID: PMC2777509
- DOI: 10.1371/journal.pone.0008030
Identification and expression analysis of ras gene in silkworm, Bombyx mori
Abstract
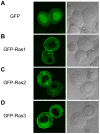
Ras proteins play important roles in development especially for cell proliferation and differentiation in various organisms. However, their functions in the most insect species are still not clear. We identified three ras cDNAs from the silk worm, Bombyx mori. These sequences corresponded to three Ras of Drosophila melanogaster, but not to three mammalian Ras (H-Ras, K-Ras, N-Ras). Subsequently, the expression profiles of ras were investigated by quantitative real-time PCR using whole body of individuals from the embryonic to adult stages, and various tissues of 4th and 5th instar larvae. Each of three Bombyx ras showed different expression patterns. We also showed membrane localization of their products. These results indicate that the three Bombyx Ras are functional and have different roles.
Conflict of interest statement
Figures


Similar articles
-
Molecular cloning and characterization of Bombyx mori CREB gene.Arch Insect Biochem Physiol. 2009 May;71(1):31-44. doi: 10.1002/arch.20292. Arch Insect Biochem Physiol. 2009. PMID: 19194985
-
Identification and characterization of Polycomb group genes in the silkworm, Bombyx mori.Mol Biol Rep. 2012 May;39(5):5575-88. doi: 10.1007/s11033-011-1362-5. Epub 2011 Dec 21. Mol Biol Rep. 2012. PMID: 22187347
-
Expression analysis and tissue distribution of two 14-3-3 proteins in silkworm (Bombyx mori).Biochim Biophys Acta. 2007 Dec;1770(12):1598-604. doi: 10.1016/j.bbagen.2007.08.005. Epub 2007 Aug 23. Biochim Biophys Acta. 2007. PMID: 17949913
-
Molecular cloning, characterization and expression analysis of ATG1 in the silkworm, Bombyx mori.Gene. 2012 Dec 15;511(2):326-37. doi: 10.1016/j.gene.2012.09.086. Epub 2012 Oct 3. Gene. 2012. PMID: 23041082
-
Structure and expression of the gene encoding a Broad-Complex homolog in the silkworm, Bombyx mori.Gene. 2004 Sep 15;339:161-72. doi: 10.1016/j.gene.2004.06.039. Gene. 2004. PMID: 15363856
Cited by
-
Transcriptomics and identification of the chemoreceptor superfamily of the pupal parasitoid of the oriental fruit fly, Spalangia endius Walker (Hymenoptera: Pteromalidae).PLoS One. 2014 Feb 5;9(2):e87800. doi: 10.1371/journal.pone.0087800. eCollection 2014. PLoS One. 2014. PMID: 24505315 Free PMC article.
-
Identification of a new Sprouty protein responsible for the inhibition of the Bombyx mori nucleopolyhedrovirus reproduction.PLoS One. 2014 Jun 10;9(6):e99200. doi: 10.1371/journal.pone.0099200. eCollection 2014. PLoS One. 2014. PMID: 24915434 Free PMC article.
-
Expression, Purification, and Characterization of Ras Protein (BmRas1) from Bombyx mori.Comp Funct Genomics. 2012;2012:747539. doi: 10.1155/2012/747539. Epub 2012 Mar 29. Comp Funct Genomics. 2012. PMID: 22536118 Free PMC article.
-
Ras1(CA) overexpression in the posterior silk gland improves silk yield.Cell Res. 2011 Jun;21(6):934-43. doi: 10.1038/cr.2011.36. Epub 2011 Mar 15. Cell Res. 2011. PMID: 21403678 Free PMC article.
-
Transcriptomics of the bed bug (Cimex lectularius).PLoS One. 2011 Jan 19;6(1):e16336. doi: 10.1371/journal.pone.0016336. PLoS One. 2011. PMID: 21283830 Free PMC article.
References
-
- Harvey JJ. An Unidentified Virus Which Causes the Rapid Production of Tumours in Mice. Nature. 1964;204:1104–1105. - PubMed
-
- Kirsten WH, Mayer LA. Morphologic responses to a murine erythroblastosis virus. J Natl Cancer Inst. 1967;39:311–335. - PubMed
-
- Marshall CJ, Hall A, Weiss RA. A transforming gene present in human sarcoma cell lines. Nature. 1982;299:171–173. - PubMed
-
- Shih TY, Papageorge AG, Stokes PE, Weeks MO, Scolnick EM. Guanine nucleotide-binding and autophosphorylating activities associated with the p21src protein of Harvey murine sarcoma virus. Nature. 1980;287:686–691. - PubMed
-
- Reuther GW, Der CJ. The Ras branch of small GTPases: Ras family members don't fall far from the tree. Curr Opin Cell Biol. 2000;12:157–165. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

