Mechanisms of the penetration of blood-borne substances into the brain
- PMID: 19949573
- PMCID: PMC2730006
- DOI: 10.2174/157015909788848901
Mechanisms of the penetration of blood-borne substances into the brain
Abstract
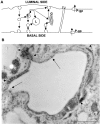
The blood-brain barrier (BBB) impedes the influx of intravascular compounds from the blood to the brain. Few blood-borne macromolecules are transferred into the brain because vesicular transcytosis in the endothelial cells is considerably limited and the tight junction is located between the endothelial cells. At the first line of the BBB, the endothelial glycocalyx which is a negatively charged, surface coat of proteoglycans, and adsorbed plasma proteins, contributes to the vasculoprotective effects of the vessels wall and are involved in maintaining vascular permeability. In the endothelial cytoplasm of cerebral capillaries, there is an asymmetrical array of metabolic enzymes such as alkaline phosphatase, acid phosphatase, 5'-nucleotidase, adenosine triphosphatase, and nucleoside diphosphatase and these enzymes contribute to inactivation of substrates. In addition, there are several types of influx or efflux transporters at the BBB, such as P-glycoprotein (P-gp), multidrug resistance associated protein, breast cancer resistance protein, organic anion transporters, organic cation transporters, organic cation transporter novel type transporters, and monocarboxylic acid transporters. P-gp, energy-dependent efflux transporter protein, is instrumental to the barrier function. Several findings recently reported indicate that endothelial P-gp contributes to efflux of undesirable substances such as beta-amyloid protein from the brain or periarterial interstitial fluid, while P-gp likely plays a crucial role in the genesis of multiple vascular abnormalities that accompany hypertension. In this review, influx and efflux mechanisms of drugs at the BBB are also reviewed and how medicines pass the BBB to reach the brain parenchyma is discussed.
Keywords: Blood-brain barrier; P-glycoprotein; tight junction..
Figures

References
-
- Bart J, Groen HJM, Hendrikse NH, van der Graaf WTA, Vaalburg W, de Vries EGE. The blood-brain barrier and oncology: new insights into function and modulation. Cancer Treat. Rev. 2000;26:449–462. - PubMed
-
- Bauer B, Hartz AWS, Fricker G, Miller DS. Modulation of P-glycoprotein transport function at the blood-brain barrier. Exp. Biol Med. 2005;230:118–127. - PubMed
-
- Bendayan R, Lee G, Bendayan M. Functional expression and localization of P-glycoprotein at the blood brain barrier. Microsc Res Tech. 2002;57:365–380. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
