Clathrin mediates integrin endocytosis for focal adhesion disassembly in migrating cells
- PMID: 19951918
- PMCID: PMC2806590
- DOI: 10.1083/jcb.200904054
Clathrin mediates integrin endocytosis for focal adhesion disassembly in migrating cells
Abstract
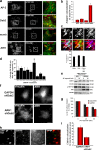
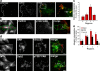
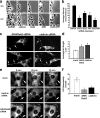
Focal adhesion disassembly is regulated by microtubules (MTs) through an unknown mechanism that involves dynamin. To test whether endocytosis may be involved, we interfered with the function of clathrin or its adaptors autosomal recessive hypercholesteremia (ARH) and Dab2 (Disabled-2) and found that both treatments prevented MT-induced focal adhesion disassembly. Surface labeling experiments showed that integrin was endocytosed in an extracellular matrix-, clathrin-, and ARH- and Dab2-dependent manner before entering Rab5 endosomes. Clathrin colocalized with a subset of focal adhesions in an ARH- and Dab2-dependent fashion. Direct imaging showed that clathrin rapidly accumulated on focal adhesions during MT-stimulated disassembly and departed from focal adhesions with integrin upon their disassembly. In migrating cells, depletion of clathrin or Dab2 and ARH inhibited focal adhesion disassembly and decreased the rate of migration. These results show that focal adhesion disassembly occurs through a targeted mechanism involving MTs, clathrin, and specific clathrin adaptors and that direct endocytosis of integrins from focal adhesions mediates their disassembly in migrating cells.
Figures
Similar articles
-
Focal adhesion disassembly requires clathrin-dependent endocytosis of integrins.FEBS Lett. 2009 Apr 17;583(8):1337-43. doi: 10.1016/j.febslet.2009.03.037. Epub 2009 Mar 22. FEBS Lett. 2009. PMID: 19306879 Free PMC article.
-
Type I phosphatidylinositol phosphate kinase beta regulates focal adhesion disassembly by promoting beta1 integrin endocytosis.Mol Cell Biol. 2010 Sep;30(18):4463-79. doi: 10.1128/MCB.01207-09. Epub 2010 Jul 12. Mol Cell Biol. 2010. PMID: 20624912 Free PMC article.
-
FAK, talin and PIPKIγ regulate endocytosed integrin activation to polarize focal adhesion assembly.Nat Cell Biol. 2016 May;18(5):491-503. doi: 10.1038/ncb3333. Epub 2016 Apr 4. Nat Cell Biol. 2016. PMID: 27043085
-
Focal adhesions, reticular adhesions, flat clathrin lattices: what divides them, what unites them?Am J Physiol Cell Physiol. 2025 Jan 1;328(1):C288-C302. doi: 10.1152/ajpcell.00821.2024. Epub 2024 Dec 9. Am J Physiol Cell Physiol. 2025. PMID: 39652817 Review.
-
Targeting and transport: how microtubules control focal adhesion dynamics.J Cell Biol. 2012 Aug 20;198(4):481-9. doi: 10.1083/jcb.201206050. J Cell Biol. 2012. PMID: 22908306 Free PMC article. Review.
Cited by
-
Targeting receptor-mediated endocytotic pathways with nanoparticles: rationale and advances.Adv Drug Deliv Rev. 2013 Jan;65(1):121-38. doi: 10.1016/j.addr.2012.09.041. Epub 2012 Sep 29. Adv Drug Deliv Rev. 2013. PMID: 23026636 Free PMC article. Review.
-
Rab5 activation promotes focal adhesion disassembly, migration and invasiveness in tumor cells.J Cell Sci. 2013 Sep 1;126(Pt 17):3835-47. doi: 10.1242/jcs.119727. Epub 2013 Jun 26. J Cell Sci. 2013. PMID: 23813952 Free PMC article.
-
Disruption of the coxsackievirus and adenovirus receptor-homodimeric interaction triggers lipid microdomain- and dynamin-dependent endocytosis and lysosomal targeting.J Biol Chem. 2014 Jan 10;289(2):680-95. doi: 10.1074/jbc.M113.518365. Epub 2013 Nov 22. J Biol Chem. 2014. PMID: 24273169 Free PMC article.
-
Integrin cytoplasmic tail interactions.Biochemistry. 2014 Feb 11;53(5):810-20. doi: 10.1021/bi401596q. Epub 2014 Jan 27. Biochemistry. 2014. PMID: 24467163 Free PMC article. Review.
-
Emerging Roles of the Endoplasmic Reticulum Associated Unfolded Protein Response in Cancer Cell Migration and Invasion.Cancers (Basel). 2019 May 6;11(5):631. doi: 10.3390/cancers11050631. Cancers (Basel). 2019. PMID: 31064137 Free PMC article. Review.
References
-
- Abercrombie M. 1980. The crawling behavior of metazoan cells. Proc. R. Soc. Lond. B. Biol. Sci. 207:129–147 10.1098/rspb.1980.0017 - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

