PYK2 interacts with MyD88 and regulates MyD88-mediated NF-kappaB activation in macrophages
- PMID: 19955209
- PMCID: PMC2830122
- DOI: 10.1189/jlb.0309125
PYK2 interacts with MyD88 and regulates MyD88-mediated NF-kappaB activation in macrophages
Abstract
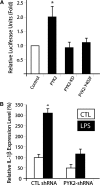
PYK2, a major cell adhesion-activated tyrosine kinase, is highly expressed in macrophages and implicated in macrophage activation and inflammatory response. However, mechanisms by which PYK2 regulates inflammatory response are beginning to be understood. In this study, we demonstrate that PYK2 interacts with MyD88, a crucial signaling adaptor protein in LPS and PGN-induced NF-kappaB activation, in vitro and in macrophages. This interaction, increased in macrophages, stimulated by LPS, requires the death domain of MyD88. PYK2-deficient macrophages exhibit reduced phosphorylation and degradation of IkappaB, an inhibitor of NF-kappaB nuclear translocation, and decreased NF-kappaB activation and IL-1beta expression by LPS. These results suggest that via interaction with MyD88, PYK2 is involved in modulating cytokine (e.g., LPS) stimulation of NF-kappaB activity and signaling, providing a mechanism underlying PYK2 regulation of an inflammatory response.
Figures




Similar articles
-
Inhibitory effect of chroman carboxamide on interleukin-6 expression in response to lipopolysaccharide by preventing nuclear factor-kappaB activation in macrophages.Eur J Pharmacol. 2006 Aug 14;543(1-3):158-65. doi: 10.1016/j.ejphar.2006.05.042. Epub 2006 Jun 2. Eur J Pharmacol. 2006. PMID: 16797005
-
Quercetin disrupts tyrosine-phosphorylated phosphatidylinositol 3-kinase and myeloid differentiation factor-88 association, and inhibits MAPK/AP-1 and IKK/NF-κB-induced inflammatory mediators production in RAW 264.7 cells.Immunobiology. 2013 Dec;218(12):1452-67. doi: 10.1016/j.imbio.2013.04.019. Epub 2013 May 9. Immunobiology. 2013. PMID: 23735482
-
Protein kinase Cdelta and protein tyrosine kinase regulate peptidoglycan-induced nuclear factor-kappaB activation and inducible nitric oxide synthase expression in mouse peritoneal macrophages in vitro.Mol Immunol. 2010 Jan;47(4):861-70. doi: 10.1016/j.molimm.2009.10.029. Epub 2009 Nov 22. Mol Immunol. 2010. PMID: 19931912
-
MD-2 regulates LPS-induced NLRP3 inflammasome activation and IL-1beta secretion by a MyD88/NF-κB-dependent pathway in alveolar macrophages cell line.Mol Immunol. 2017 Oct;90:1-10. doi: 10.1016/j.molimm.2017.06.035. Epub 2017 Jun 24. Mol Immunol. 2017. PMID: 28654770
-
LPS-induced MCP-1 expression in human microvascular endothelial cells is mediated by the tyrosine kinase, Pyk2 via the p38 MAPK/NF-kappaB-dependent pathway.Mol Immunol. 2009 Feb;46(5):962-8. doi: 10.1016/j.molimm.2008.09.022. Epub 2008 Oct 26. Mol Immunol. 2009. PMID: 18954908 Free PMC article.
Cited by
-
Enhanced phagocytosis associated with multinucleated microglia via Pyk2 inhibition in an acute β-amyloid infusion model.J Neuroinflammation. 2024 Aug 6;21(1):196. doi: 10.1186/s12974-024-03192-7. J Neuroinflammation. 2024. PMID: 39107821 Free PMC article.
-
Calcium/calmodulin-dependent protein kinase kinase 2 regulates macrophage-mediated inflammatory responses.J Biol Chem. 2012 Mar 30;287(14):11579-91. doi: 10.1074/jbc.M111.336032. Epub 2012 Feb 14. J Biol Chem. 2012. PMID: 22334678 Free PMC article.
-
L-plastin enhances NLRP3 inflammasome assembly and bleomycin-induced lung fibrosis.Cell Rep. 2022 Mar 15;38(11):110507. doi: 10.1016/j.celrep.2022.110507. Cell Rep. 2022. PMID: 35294888 Free PMC article.
-
Mycobacterial phosphatidylinositol mannoside 6 (PIM6) up-regulates TCR-triggered HIV-1 replication in CD4+ T cells.PLoS One. 2013 Nov 25;8(11):e80938. doi: 10.1371/journal.pone.0080938. eCollection 2013. PLoS One. 2013. PMID: 24282561 Free PMC article.
-
Molecular mechanisms of regulation of Toll-like receptor signaling.J Leukoc Biol. 2016 Nov;100(5):927-941. doi: 10.1189/jlb.2MR0316-117RR. Epub 2016 Jun 24. J Leukoc Biol. 2016. PMID: 27343013 Free PMC article. Review.
References
-
- Li X, Qin J. Modulation of Toll-interleukin 1 receptor mediated signaling. J Mol Med. 2005;83:258–266. - PubMed
-
- Davidson D J, Currie A J, Bowdish D M, Brown K L, Rosenberger C M, Ma R C, Bylund J, Campsall P A, Puel A, Picard C, Casanova J L, Turvey S E, Hancock R E, Devon R S, Speert D P. IRAK-4 mutation (Q293X): rapid detection and characterization of defective post-transcriptional TLR/IL-1R responses in human myeloid and non-myeloid cells. J Immunol. 2006;177:8202–8211. - PMC - PubMed
-
- O'Neill L A, Bowie A G. The family of five: TIR-domain-containing adaptors in Toll-like receptor signaling. Nat Rev Immunol. 2007;7:353–364. - PubMed
-
- Rakoff-Nahoum S, Medzhitov R. Toll-like receptors and cancer. Nat Rev Cancer. 2009;9:57–63. - PubMed
-
- Moynagh P N. The Pellino family: IRAK E3 ligases with emerging roles in innate immune signaling. Trends Immunol. 2009;30:33–42. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

