Positive regulatory control loop between gut leptin and intestinal GLUT2/GLUT5 transporters links to hepatic metabolic functions in rodents
- PMID: 19956534
- PMCID: PMC2780353
- DOI: 10.1371/journal.pone.0007935
Positive regulatory control loop between gut leptin and intestinal GLUT2/GLUT5 transporters links to hepatic metabolic functions in rodents
Abstract
Background and aims: The small intestine is the major site of absorption of dietary sugars. The rate at which they enter and exit the intestine has a major effect on blood glucose homeostasis. In this study, we determine the effects of luminal leptin on activity/expression of GLUT2 and GLUT5 transporters in response to sugars intake and analyse their physiological consequences.
Methodology: Wistar rats, wild type and AMPKalpha(2) (-/-) mice were used. In vitro and in vivo isolated jejunal loops were used to quantify transport of fructose and galactose in the absence and the presence of leptin. The effects of fructose and galactose on gastric leptin release were determined. The effects of leptin given orally without or with fructose were determined on the expression of GLUT2/5, on some gluconeogenesis and lipogenic enzymes in the intestine and the liver.
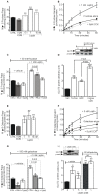
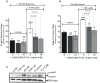
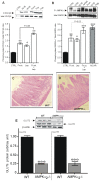
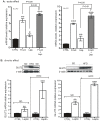
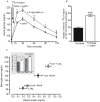
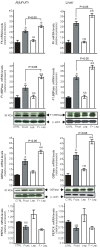
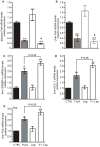
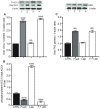
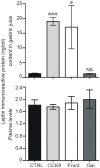
Principal findings: First, in vitro luminal leptin activating its receptors coupled to PKCbetaII and AMPKalpha, increased insertion of GLUT2/5 into the brush-border membrane leading to enhanced galactose and fructose transport. Second in vivo, oral fructose but not galactose induced in mice a rapid and potent release of gastric leptin in gastric juice without significant changes in plasma leptin levels. Moreover, leptin given orally at a dose reproducing comparable levels to those induced by fructose, stimulated GLUT5-fructose transport, and potentiated fructose-induced: i) increase in blood glucose and mRNA levels of key gluconeogenesis enzymes; ii) increase in blood triglycerides and reduction of mRNA levels of intestinal and hepatic Fasting-induced adipocyte factor (Fiaf) and iii) increase in SREBP-1c, ACC-1, FAS mRNA levels and dephosphorylation/activation of ACC-1 in liver.
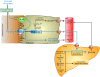
Conclusion/significance: These data identify for the first time a positive regulatory control loop between gut leptin and fructose in which fructose triggers release of gastric leptin which, in turn, up-regulates GLUT5 and concurrently modulates metabolic functions in the liver. This loop appears to be a new mechanism (possibly pathogenic) by which fructose consumption rapidly becomes highly lipogenic and deleterious.
Conflict of interest statement
Figures
References
-
- Kellett GL, Brot-Laroche E. Apical GLUT2: a major pathway of intestinal sugar absorption. Diabetes. 2005;54:3056–3062. - PubMed
-
- Ishikawa Y, Eguchi T, Ishida H. Mechanism of beta-adrenergic agonist-induced transmural transport of glucose in rat small intestine. Regulation of phosphorylation of SGLT1 controls the function. Biochim Biophys Acta. 1997;1357:306–318. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

