Direct activation of RhoA by reactive oxygen species requires a redox-sensitive motif
- PMID: 19956681
- PMCID: PMC2778012
- DOI: 10.1371/journal.pone.0008045
Direct activation of RhoA by reactive oxygen species requires a redox-sensitive motif
Abstract
Background: Rho family GTPases are critical regulators of the cytoskeleton and affect cell migration, cell-cell adhesion, and cell-matrix adhesion. As with all GTPases, their activity is determined by their guanine nucleotide-bound state. Understanding how Rho proteins are activated and inactivated has largely focused on regulatory proteins such as guanine nucleotide exchange factors (GEFs) and GTPase activating proteins (GAPs). However, recent in vitro studies have indicated that GTPases may also be directly regulated by redox agents. We hypothesized that this redox-based mechanism occurs in cells and affects cytoskeletal dynamics, and in this report we conclude this is indeed a novel mechanism of regulating the GTPase RhoA.
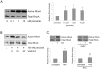
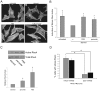
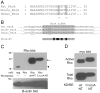
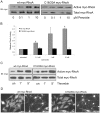
Methodology/principal findings: In this report, we show that RhoA can be directly activated by reactive oxygen species (ROS) in cells, and that this requires two critical cysteine residues located in a unique redox-sensitive motif within the phosphoryl binding loop. First, we show that ROS can reversibly activate RhoA and induce stress fiber formation, a well characterized readout of RhoA activity. To determine the role of cysteine residues in this mechanism of regulation, we generated cysteine to alanine RhoA mutants. Mutation of these cysteines abolishes ROS-mediated activation and stress fiber formation, indicating that these residues are critical for redox-regulation of RhoA. Importantly, these mutants maintain the ability to be activated by GEFs.
Conclusions/significance: Our findings identify a novel mechanism for the regulation of RhoA in cells by ROS, which is independent of classical regulatory proteins. This mechanism of regulation may be particularly relevant in pathological conditions where ROS are generated and the cellular redox-balance altered, such as in asthma and ischemia-reperfusion injury.
Conflict of interest statement
Figures
References
-
- Nobes CD, Hall A. Rho, rac, and cdc42 GTPases regulate the assembly of multimolecular focal complexes associated with actin stress fibers, lamellipodia, and filopodia. Cell. 1995;81:53–62. - PubMed
-
- Ridley AJ, Hall A. The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell. 1992;70:389–399. - PubMed
-
- Ridley AJ, Paterson HF, Johnston CL, Diekmann D, Hall A. The small GTP-binding protein rac regulates growth factor-induced membrane ruffling. Cell. 1992;70:401–410. - PubMed
-
- Burridge K, Wennerberg K. Rho and Rac take center stage. Cell. 2004;116:167–179. - PubMed
-
- DerMardirossian C, Bokoch GM. GDIs: central regulatory molecules in Rho GTPase activation. Trends Cell Biol. 2005;15:356–363. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

