Ror1-Ror2 complexes modulate synapse formation in hippocampal neurons
- PMID: 19958813
- PMCID: PMC2814931
- DOI: 10.1016/j.neuroscience.2009.11.056
Ror1-Ror2 complexes modulate synapse formation in hippocampal neurons
Abstract
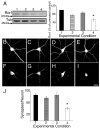
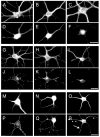
Ror1 and Ror2, a small family of tyrosine kinase receptors, have been implicated in multiple aspects of brain development in C. elegans and X. laevis. More recently, we have shown that these receptors modulate the rate of neurite elongation in cultured rat hippocampal neurons. However, no information is available regarding a potential role of these receptors in other developmental milestones in mammalian central neurons. Neither is the identity known of the Ror ligand(s) and/or the signal transduction pathway(s) in which they participate. Here we report that the down regulation of either Ror1 or Ror2 led to a significant decrease in synapse formation in cultured hippocampal neurons. Simultaneous targeting of Ror proteins, however, did not result in an additive phenotype. Our results also indicated that Ror1 and Ror2 physically interact in the mouse brain, suggesting that they might function as heterodimers in central neurons. In addition, these Ror complexes interacted with Wnt-5a mediating its effects on synaptogenesis. Together, these data suggest that Ror proteins play a key role in Wnt-5a-activated signaling pathways leading to synapse formation in the mammalian CNS.
Figures





Similar articles
-
Ror family receptor tyrosine kinases regulate the maintenance of neural progenitor cells in the developing neocortex.J Cell Sci. 2012 Apr 15;125(Pt 8):2017-29. doi: 10.1242/jcs.097782. Epub 2012 Feb 10. J Cell Sci. 2012. PMID: 22328498
-
The ROR2 tyrosine kinase receptor regulates dendritic spine morphogenesis in hippocampal neurons.Mol Cell Neurosci. 2015 Jul;67:22-30. doi: 10.1016/j.mcn.2015.05.002. Epub 2015 May 21. Mol Cell Neurosci. 2015. PMID: 26003414
-
Role of the Ror family receptors in Wnt5a signaling.In Vitro Cell Dev Biol Anim. 2024 May;60(5):489-501. doi: 10.1007/s11626-024-00885-4. Epub 2024 Apr 8. In Vitro Cell Dev Biol Anim. 2024. PMID: 38587578 Review.
-
Diverse roles for the ror-family receptor tyrosine kinases in neurons and glial cells during development and repair of the nervous system.Dev Dyn. 2018 Jan;247(1):24-32. doi: 10.1002/dvdy.24515. Epub 2017 Jun 1. Dev Dyn. 2018. PMID: 28470690 Review.
-
RoR2 functions as a noncanonical Wnt receptor that regulates NMDAR-mediated synaptic transmission.Proc Natl Acad Sci U S A. 2015 Apr 14;112(15):4797-802. doi: 10.1073/pnas.1417053112. Epub 2015 Mar 30. Proc Natl Acad Sci U S A. 2015. PMID: 25825749 Free PMC article.
Cited by
-
The RTK Interactome: Overview and Perspective on RTK Heterointeractions.Chem Rev. 2019 May 8;119(9):5881-5921. doi: 10.1021/acs.chemrev.8b00467. Epub 2018 Dec 27. Chem Rev. 2019. PMID: 30589534 Free PMC article. Review.
-
Canonical and noncanonical Wnt signaling in neural stem/progenitor cells.Cell Mol Life Sci. 2015 Nov;72(21):4157-72. doi: 10.1007/s00018-015-2028-6. Epub 2015 Aug 26. Cell Mol Life Sci. 2015. PMID: 26306936 Free PMC article. Review.
-
Neurodevelopmental Perspectives on Wnt Signaling in Psychiatry.Mol Neuropsychiatry. 2017 Feb;2(4):219-246. doi: 10.1159/000453266. Epub 2017 Jan 13. Mol Neuropsychiatry. 2017. PMID: 28277568 Free PMC article. Review.
-
A multiomic atlas of the aging hippocampus reveals molecular changes in response to environmental enrichment.Nat Commun. 2024 Jul 16;15(1):5829. doi: 10.1038/s41467-024-49608-z. Nat Commun. 2024. PMID: 39013876 Free PMC article.
-
Distinct Patterns of Stromal and Tumor Expression of ROR1 and ROR2 in Histological Subtypes of Epithelial Ovarian Cancer.Transl Oncol. 2017 Jun;10(3):346-356. doi: 10.1016/j.tranon.2017.01.014. Epub 2017 Mar 23. Transl Oncol. 2017. PMID: 28342318 Free PMC article.
References
-
- Al-Shawi R, Ashton SV, Underwood C, Simons JP. Expression of the Ror1 and Ror2 receptor tyrosine kinase genes during mouse development. Dev Genes Evol. 2001;211:161–171. - PubMed
-
- Bhanot P, Brink M, Samos CH, Hsieh JC, Wang Y, Macke JP, Andrew D, Nathans J, Nusse R. A new member of the frizzled family from Drosophila functions as a Wingless receptor. Nature. 1996;382:225–230. - PubMed
-
- Billiard J, Way DS, Seestaller-Wehr LM, Moran RA, Mangine A, Bodine PV. The Orphan Receptor Tyrosine Kinase Ror2 Modulates Canonical Wnt Signaling in Osteoblastic Cells. Mol Endocrinol. 2005;19:90–101. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

